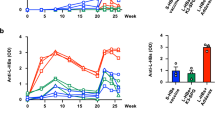
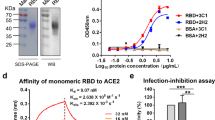
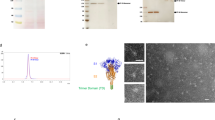
Abstract
The worldwide importance of human hepatitis B virus infection and the toll it takes in chronic liver disease, cirrhosis and hepatocarcinoma, make it imperative that a vaccine be developed for worldwide application1. Human hepatitis B vaccines2–6 are presently prepared using hepatitis B surface antigen (HBsAg) that is purified from the plasma of human carriers of hepatitis B virus infection. The preparation of hepatitis B vaccine from a human source is restricted by the available supply of infected human plasma and by the need to apply stringent processes that purify the antigen and render it free of infectious hepatitis B virus and other possible living agents that might be present in the plasma. Joint efforts between our laboratories and those of Drs W. Rutter and B. Hall led to the preparation of vectors carrying the DNA sequence7,8 for HBsAg and antigen expression in the yeast Saccharomyces cerevisiae9. Here we describe the development of hepatitis B vaccine of yeast cell origin. HBsAg of subtype adw was produced in recombinant yeast cell culture, and the purified antigen in alum formulation stimulated production of antibody in mice, grivet monkeys and chimpanzees. Vaccinated chimpanzees were totally protected when challenged intravenously with either homologous or heterologous subtype adr and ayw virus of human serum source. This is the first example of a vaccine produced from recombinant cells which is effective against a human viral infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Deinhardt, F. & Gust, I. D. Bull. Wld Hlth Org. 60, 661–691 (1982).
Hilleman, M. R. et al. Viral Hepatitis (eds Vyas, G. N., Cohen, S. N. & Schmid, R.) 525–537 (The Franklin Institute Press, Philadelphia, 1978).
Hilleman, M. R. et al. in Viral Hepatitis 1981 int. Symp. (eds Szmuness, W., Alter, H. & Maynard, J.) 385–397 (The Franklin Institute Press, Philadelphia, 1982).
Buynak, E. B. et al. J. Am. med. Ass. 235, 2832–2834 (1976).
Adamowicz, P. et al. in INSERM Symp. No. 18 (eds Maupas, P. & Guesry, P.) 37–49 (Elsevier, Amsterdam, 1981).
Coutinho, R. A. et al. Br. med. J. 286, 1305–1308 (1983).
Valenzuela, P. et al. Nature 280, 815–819 (1979).
Edman, J. C., Hallewell, R. A., Valenzuela, P., Goodman, H. M. & Rutter, W. J. Nature 291, 503–506 (1981).
Valenzuela, P., Medina, A., Rutter, W. J., Ammerer, G. & Hall, B. D. Nature 298, 347–350 (1982).
Alexander, J. J., Bey, E. M., Geddes, E. W. & Lecatsas, G. S. Afr. med. J. 50, 2124–2128 (1976).
Barin, F., Maupas, P., Coursaget, P., Goudeau, A. & Chiron, J. P. in INSERM Symp. No. 18 (eds Maupas, P. & Guesry, P.) 263–266 (Elsevier, Amsterdam, 1981).
Moriarty, A. M., Hoyer, B. H., Shih, J. W-K., Gerin, J. L. & Hamer, D. H. Proc. natn. Acad. Sci. U.S.A. 78, 2606–2610 (1981).
Dubois, M. F., Pourcel, C., Rousset, S., Chany, C. & Tiollais, P. Proc. natn. Acad. Sci. U.S.A. 77, 4549–4553 (1980).
Burrell, J. C., Mackay, P., Greenway, P. J., Hofschneider, P. H. & Murray, K. Nature 279, 43–47 (1979).
Lerner, R. A. et al. Proc. natn. Acad. Sci. U.S.A. 78, 3403–3407 (1981).
Dreesman, G. R. et al. Nature 295, 158–160 (1982).
Bhatnagar, P. K. et al. Proc. natn. Acad. Sci. U.S.A. 79, 4400–4404 (1982).
Prince, A. M., Ikram, H. & Hopp, T. P. Proc. natn. Acad. Sci. U.S.A. 79, 579–582 (1982).
Gerin, J. L. et al. Proc. natn. Acad. Sci. U.S.A. 80, 2365–2369 (1983).
Smith, G. L., Mackett, M. & Moss, B. Nature 302, 490–495 (1983).
Miyanohara, A. et al. Proc. natn. Acad. Sci. U.S.A. 80, 1–5 (1983).
Hitzeman, R. A. et al. Nucleic Acids Res. 11, 2745–2763 (1983).
Deters, D., Muller, U. & Homberger, H. Analyt. Biochem. 70, 263–267 (1976).
Ono, Y. et al. Nucleic Acids Res. 11, 1747–1757 (1983).
Gerety, R. J., Tabor, E., Purcell, R. H. & Tyeryar, F. J. J. infect. Dis. 140, 642–648 (1979).
Szmuness, W., Stevens, C. E., Zang, E. A., Harley, E. J. & Kellner, A. Hepatology 1, 377–385 (1981).
Francis, D. P. et al. Ann. intern. Med. 97, 362–366 (1982).
Szmuness, W. et al. New Engl. J. Med. 307, 1481–1486 (1982).
Lowry, O. H., Rosebrough, N. J., Farr, A. L. & Randall, R. J. biol. Chem. 193, 265–275 (1951).
Laemmli, U. K. Nature 227, 680–685 (1970).
Morrissey, J. H. Analyt. Biochem. 117, 307–310 (1981).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McAleer, W., Buynak, E., Maigetter, R. et al. Human hepatitis B vaccine from recombinant yeast. Nature 307, 178–180 (1984). https://doi.org/10.1038/307178a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/307178a0
This article is cited by
-
Intratumoural microbiota: a new frontier in cancer development and therapy
Signal Transduction and Targeted Therapy (2024)
-
Targeting aging and age-related diseases with vaccines
Nature Aging (2024)
-
Endotoxin-free gram-negative bacterium as a system for production and secretion of recombinant proteins
Applied Microbiology and Biotechnology (2023)
-
Molecular engineering improves antigen quality and enables integrated manufacturing of a trivalent subunit vaccine candidate for rotavirus
Microbial Cell Factories (2021)
-
Establishment of a yeast-based VLP platform for antigen presentation
Microbial Cell Factories (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.