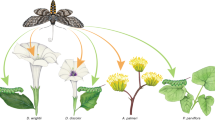
Abstract
INTERSPECIFIC mutualisms are regarded as having evolved from antagonistic or commensalistic interactions, with most mutualisms remaining facultative but some having coevolved into obligate reciprocal dependency1–4. Underlying mutualism is an intrinsic conflict between the parties, in that each is under selection for increased exploitation of the other3–7. Theoretical models suggest that this conflict is a source of evolutionary instability, and that evolution of 'cheating' by one party may lead to reciprocal extinction4,6,7. Here we present phylogenetic evidence for reversal of an obligate mutualism: within the yucca moth complex, distinct cheater species derived from obligate pollinators inflict a heavy cost on their yucca hosts by laying their eggs but not pollinating the yucca. Phylogenetic data show the cheaters to have existed for a long time. Coexisting pollinators and cheaters are not sister taxa, supporting predictions that evolution of cheating within a single pollinator is evolutionary unstable. Several lines of evidence support a hypothesis that host shifts preceded the reversal of obligate mutualism. Host or partner shifts is a mechanism that can provide a route of evolutionary escape among obligate mutualists in general.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Thompson, J. N. The Revolutionary Process (University of Chicago Press, 1994).
Gargas, A., dePriest, P. T., Grube, M. & Tehler, A. Science 268, 1492–1495 (1995).
Axelrod, R. & Hamilton, W. D. Science 242, 1385–1390 (1981).
Bull, J. J. & Rice, W. R. J. theor. Biol. 149, 63–74 (1991).
Pellmyr, O. & Huth, C. J. Nature 372, 257–260 (1994).
Trivers, R. L. Q. Rev. Biol. 46, 35–57 (1971).
Soberon Mainero, J. & Martinez del Rio, C. in The Biology of Mutualism (ed. Boucher, D. H.) 192–216 (Oxford Univ. Press, New York, 1985).
Riley, C. V. Am. Mo. bot. Gdn 3, 99–158 (1892).
Powell, J. A. Trends Ecol. Evol. 7, 10–14 (1992).
Richter, K. S. & Weis, A. E. Nature 376, 557–558 (1995).
Keeley, J. E., Keeley, S. C. & Ikeda, D. A. Am. Midl. Nat. 115, 1–9 (1986).
Pellmyr, O. & Thompson, J. N. Proc. natn. Acad. Sci. U.S.A. 89, 2927–2929 (1992).
Davis, D. R. U.S. natn. Mus. Bull. 255, 1–170 (1967).
Miles, N. J. J. Lepid. Soc. 37, 207–216 (1983).
Tyre, A. J. & Addicott, J. F. Oecologia 94, 173–175 (1993).
Riley, C. V. Proc. Am. Ass. Advmt Sci. 29, 617–639 (1881).
Feist, M. A. thesis, Univ. Cincinnati (1995).
Felsenstein, J. Phylogenetic Inference Package (PHYLIP) version 3.5c (University of Washington, 1993).
Kimura, M. J. molec. Evol. 16, 111–120 (1980).
Wright, S. The Theory of Gene Frequencies Vol. 2 (Univ. of Chicago Press, 1969).
Dawkins, R. The Selfish Gene, 2nd edn (Oxford Univ. Press, 1989).
Vickery, W. L., Giraldeau, L.-C., Templeton, J. J., Kramer, D. L. & Chapman, C. A. Am. Nat. 137, 847–863 (1991).
Yamauchi, A. Am. Nat. 145, 434–456 (1995).
Galil, J. & Eisikowitch, D. Tijdschr. Ent. 112, 1–13 (1969).
Wiebes, J. T. Proc. K. ned. Akad. Wet. 97, 491–495 (1994).
Kishino, H. & Hasegawa, M. J. molec. Evol. 29, 170–179 (1989).
Brown, J. L., Pellmyr, O., Thompson, J. N. & Harrison, R. G. Ann. ent. Soc. Am. 87, 795–802.
McKelvey, S. D. Yuccas of the Southwestern United States (Arnold Arboretum, Jamaica Plain, MA, 1938, 1947).
Swofford, D. L. Phylogenetic Analysis Using Parsimony, version 3.1 (Illinois Natural History Survey, Champaign, Urbana, IL, 1993).
Clary, D. O. & Wolstenholme, D. R. J. molec. Evol. 22, 252–571 (1985).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pellmyr, O., Leebens-Mack, J. & Huth, C. Non-mutualistic yucca moths and their evolutionary consequences. Nature 380, 155–156 (1996). https://doi.org/10.1038/380155a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/380155a0
This article is cited by
-
Pollinator effectiveness is affected by intraindividual behavioral variation
Oecologia (2021)
-
Foundress numbers and the timing of selective events during interactions between figs and fig wasps
Scientific Reports (2019)
-
Two’s company, three’s a crowd: co-occurring pollinators and parasite species in Breynia oblongifolia (Phyllanthaceae)
BMC Evolutionary Biology (2018)
-
Plant reproductive traits mediate tritrophic feedback effects within an obligate brood-site pollination mutualism
Oecologia (2015)
-
Asymmetric or diffusive co-evolution generates meta-populations in fig-fig wasp mutualisms
Science China Life Sciences (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.