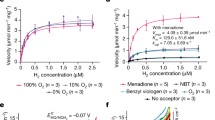
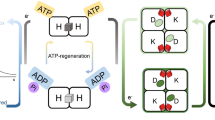
Abstract
Wilson and Burris1 reviewed the evidence implicating hydrogenase in biological nitrogen fixation and suggested two working hypotheses for the mechanism of nitrogen fixation which included the participation of hydrogenase. The relation between hydrogenase and nitrogen fixation was strengthened further when Kamen and Gest2 discovered Rhodospirillum rubrum to be a nitrogen fixer and found that nitrogen, either as such or as ammonium, inhibited its photoproduction of hydrogen. Purified hydrogenase preparations from Clostridium pasteurianum have shown spectral changes when exposed to nitrogen relative to spectra under hydrogen, oxygen, nitric oxide or a vacuum3. Although the initial report4 of hydrogenase in root-nodule bacteria was not verified by later, more extensive tests5 with nodules and bacteria from nodules, Wilson6 recently has reported that a preparation of disintegrated soy-bean nodules exhibited spectral shifts in the presence, as compared to the absence, of hydrogen, suggestive of the activity of a hydrogenase. The absence of a clear demonstration of hydrogenase in nodules often has been cited in discussion of the comparative biochemistry of nitrogen fixation as an irregularity. Evidence from the present communication removes this irregularity and establishes a further point of uniformity among nitrogen-fixing agents.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wilson, P. W., and Burris, R. H., Bact. Rev., 11, 41 (1947).
Kamen, Martin D., and Gest, Howard, Science, 109, 560 (1949).
Shug, A. L., Hamilton, P. B., and Wilson, P. W., “Inorganic Nitrogen Metabolism”, 344 edit. by McElroy, W. D., and Glass, B. (Johns Hopkins Press, Baltimore, Maryland, 1956).
Phelps, A. S., and Wilson, P. W., Proc. Soc. Exp. Biol. and Med., 47, 473 (1941).
Wilson, P. W., Burris, R. H., and Coffee, W. B., J. Biol. Chem, 147, 475 (1943).
Wilson, P. W., Science, 123, 676 (1956).
Hoagland, D. R., and Arnon, D. I., University of California Agricultural Station Circular 347 (1938).
Burris, R. E., Magee, W. E., and Bach, M. K., Ann. Acad. Sci.Fennicae, Ser. A, II, 60, A. I. Vlrtanen Homage Vol., 190 (1955).
Aprison, M. H., Magee, W. E., and Burris, R. H., J. Biol. Chem., 208, 29 (1954).
Aprison, M. H., and Burris, R. H., Science, 115, 264 (1952).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
HOCH, G., LITTLE, H. & BURRIS, R. Hydrogen Evolution from Soy-Bean Root Nodules. Nature 179, 430–431 (1957). https://doi.org/10.1038/179430a0
Issue Date:
DOI: https://doi.org/10.1038/179430a0
This article is cited by
-
Hydrogen oxidation in soil following rhizobial H2 production due to N2 fixation by a Vicia faba-Rhizobium leguminosarum symbiosis
Biology and Fertility of Soils (1991)
-
Acetylene reduction, H2 evolution and 15N2 fixation in the Alnus incana-Frankia symbiosis
Planta (1986)
-
Hydrogen accumulation by H2-uptake negative strains of Rhizobium
Plant and Soil (1985)
-
H2-Recycling system in mungbean Rhizobium in relation to N2-fixation
Archives of Microbiology (1981)
-
Selection of symbiotically energy efficient strains of Rhizobium japonicum by their ability to induce a H2-uptake hydrogenase in the free-living state
Archives of Microbiology (1981)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.