Abstract
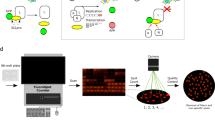
We have developed a high capacity screen for compounds that inhibit the 3C protease of human rhinovirus–1b. The assay uses a recombinant strain of Escherichia coli expressing both the protease and a tetracycline resistance–conferring protein modified to contain the minimal protease cleavage site. Cultures growing in microtiter plates containing tetracycline are treated with potential inhibitors and simultaneously monitored for change in growth over time using an oxygen probe. Most of the cultures, not containing an inhibitor of the 3C protease, show reduced growth due to cleavage of the essential gene product; normal growth is seen only in the infrequent culture that contains an inhibitor. In the present example, we have used the tetA gene of plasmid pACYC184 as the modified gene. The system has been validated using inhibitors of protease 3C, and has been used to identify three new inhibitors of the enzyme, active in the micromolar range.
Similar content being viewed by others
Article PDF
References
Scharpe, S., De Meester, I., Hendriks, D., Vanhoof, G., van Sande, M. and Vriend, G. 1991. Proteases and their inhibitors: today and tomorrow. Biochimie. 73: 121–126.
Powers, J.C.B. and Bengali, Z.H. 1987. Conference report—Elastase inhibitors for the treatment of emphysema—Approaches to synthesis and biological evaluation. J. Enzyme Inhibition. 1: 311–319.
Buo, L., Aasen, A.O., Karlsrud, T.S., Johansen, H. T. and Sivertsen, S.M. 1990. The role of proteases in growth, invasion, and spread of cancer cells. Tidsskr Nor. Laegeforen. 110: 3753–3756.
Douglas, L.J. 1988. Candida proteinases and candidosis. Crit. Rev. Biotechnol. 8: 121–129.
Lozitsky, V.P., Fedchuk, A.S., Puzis, L.E., Buiko, V.P., Girlya, Y.I. and Bubnov, V.V. 1987. Participation of the proteolysis system in realization of influenza virus virulence and development of the infectious process: Antiviral effect of protease inhibitors. Vopr. Virusol. 32: 413–419.
Walpole, C.S.J. and Wrigglesworth, R. 1989. Enzyme inhibitors in medicine. Nat. Prod. Rep. 6: 311–346.
Larson, H.E., Reed, S.E. and Tyrrell, D.A.J. 1980. Isolation of rhinoviruses and coronaviruses from 38 colds in adults. J. Med. Virol. 5: 221–229.
Fox, J.P. 1976. Is a rhinovirus vaccine possible? Amer. J. Epidemiol. 103: 345–354.
Krausslich, H.-G. and Wimmer, E. 1988. Viral proteinases. Ann. Rev. Bio-chem. 57: 701–754.
Taylor, A., Brown, D.P., Kadam, S. et al. 1992. High-level expression and purification of mature HIV-1 protease in Escherichia coli under control of the araBAD promoter. Appl. Microbiol. Biotechnol. 37: 205–210.
Chang, A.C.Y. and Cohen, S.N. 1978. Construction and characterization of amplifiable multicopy DNA cloning vehicles derived from the p15A cryptic miniplasmid. J. Bacteriol. 134: 1141–1156.
Block, T.M. and Grafstrom, R.H. 1990. Novel bacteriological assay for detection of potential antiviral agents. Antimicrob. Agents Chemother. 34: 2337–2341.
Cordingley, M.G., Register, R.B., Callahan, P.L., Garsky, V.M. and Colonno, R.J. 1989. Cleavage of small peptides in vitro by human rhinovirus 14 3C protease expressed in Escherichia coli. J. Virology. 63: 5037–5045.
Cordingley, M.G., Callahan, P.L., Sardana, V.V., Garsky, V.M. and Colonno, R.J. 1990. Substrate requirements of human rhinovirus 3C protease for peptide cleavage in vitro. J. Biol. Chem. 265: 9062–9065.
Goldberg, A.L. 1972. Degradation of abnormal proteins in Escherichia coli. Proc. Natl. Acad. Sci. USA 69: 422–426.
O'Rourke, S.V., Tanner, J.E. and Stitt, D.T. 1992. A method to identify methicillin resistant Staphylococcus aureus within 4 hours. Abstr. Ann. Meet. Am. Soc. Microbiol. 92: 462.
Payne, J.W. and Gilvarg, C. 1968. Size restriction on peptide utilization in Escherichia coli. J. Biol. Chem. 243: 6291–6299.
Merlin, T.L., Davis, G.E., Anderson, W.L., Moyzis, R.K. and Griffith, J.K. 1989. Aminoglycoside uptake increased by tet gene expression. Antimicrob. Agents Chemother. 33: 1549–1552.
Robenson, D.J. and Strobel, G.A. 1982. Deoxyradicinin, a novel phytotoxin from Altenaria helianthi. Phytochemistry. 21: 2359–2362.
Raistrick, H. and Smith, G. 1941. Antibacterial substances from mould. I. Citrinin, a metabolic product from Penicillium citrinum. Thorn. Chemistry and Industry. 1941: 828–830.
Johnson, L.E. and Dietz, A. 1968. Kalafungin, a new antibiotic produced by Streptomyces tanashiensis strain Kala. Appl. Microbiol. 16: 1815–1821.
Bergy, M.E. 1968. Kalafungin, a new broad spectrum antibiotic. Isolation and characterization. J. Antibiot. (Tokyo) 21: 454–457.
Singh, S.B., Cordingly, M.G., Ball, R.G., Smith, J.L., Dombrowski, A.W. and Goetz, M.A. 1991. Structure and stereochemistry of thysanone: a novel human rhinovirus 3C-protease inhibitor from Thysanophora penicilloides. Tetrahedron Lett. 32: 5279–5282.
Beynon, R.J. and Bond, J.S. 1989. XVIII. Proteolytic Enzymes: A Practical Approach. Practical Approach Series, IRL Press, London.
Jacobs, F.A., Romeyer, F.M., Beauchemin, M. and Brousseau, R. 1989. Human metallothionein-II is synthesized as a stable membrane-localized fusion protein in Escherichia coli. Gene. 83: 95–103.
Bertani, G. 1951. Studies on lysogenesis. I. The mode of phage liberation by lysogenic Excherichia coli. J. Bacteriol. 62: 293–300.
Zelenetz, A.D. 1992. Construction of complex directional complementary DNA libraries in SfiI. Methods Enzymol. 216: 517–530.
Maniatis, T., Fritsch, E.F. and Sambrook, J. 1989. Molecular Cloning—A Laboratory Manual. 2nd. Ed. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N. Y.
Hughes, P.J., North, C., Jellis, C.H., Minor, P.D. and Stanway, G. 1988. The nucleotide sequence of human rhinovirus IB: molecular relationships within the rhinovirus genus. J. Gen. Virol. 69: 49–58.
Merrifield, R.B. 1963. Solid phase peptide synthesis. I. The synthesis of a tetrapeptide. J. Am. Chem. Soc. 85: 2149–2154.
Watts, R.J. and Crosby, G.A. 1971. Spectroscopic characterization of complexes of ruthenium (II) and indium (III) with 4,4′-diphenyl–2,2′-bipyridine and 4,7-diphenyl–1,10-phenanthroline. J. Am. Chem. Soc. 93: 3184–3188.
Wilcox, G. 1974. The interaction of L-arabinose and D-fucose with AraC protein. J. Biol. Chem. 249: 6892–6894
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
McCall, J., Kadam, S. & Katz, L. A High Capacity Microbial Screen for Inhibitors of Human Rhinovirus Protease 3C. Nat Biotechnol 12, 1012–1016 (1994). https://doi.org/10.1038/nbt1094-1012
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt1094-1012
This article is cited by
-
Viral cysteine proteinases
Perspectives in Drug Discovery and Design (1996)
-
Protease-Dependent Streptomycin Sensitivity in E. coli—A System for Protease Inhibitor Selection
Nature Biotechnology (1995)