The compounds Ba3(PO4)2 and Sr3(PO4)2 are rhombohedral and isostructural. The unit cell contains one molecule. The dimensions of the unit rhombohedron and the calculated densities are: Ba3(PO4)2: a = 7.696 ± 0.002 kX, ![[alpha]](/logos/entities/alpha_rmgif.gif) = 42° 35' ± 2',
= 42° 35' ± 2', ![[rho]](/logos/entities/rho_rmgif.gif) = 5.25 g.cm-3; Sr3(PO4)2: a = 7.280 ± 0.002 kX,
= 5.25 g.cm-3; Sr3(PO4)2: a = 7.280 ± 0.002 kX, ![[alpha]](/logos/entities/alpha_rmgif.gif) = 43° 21' ± 2', 4.53 g.cm-3. The space group is R
= 43° 21' ± 2', 4.53 g.cm-3. The space group is R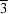 m and the atomic positions are: 1 Ba in (0, 0, 0), 2 Ba in ±(u1, u1, u1), 2 P in ±(u2, u2, u2), 2 O in ±(u3, u3, u3), 6 O in ±(x, x, z) (x, z, x) (z, x, x); with parameter values u1 = 0.208, u2 = 0.412, u3 = 0.337, x = 0.285 and z = 0.742.
m and the atomic positions are: 1 Ba in (0, 0, 0), 2 Ba in ±(u1, u1, u1), 2 P in ±(u2, u2, u2), 2 O in ±(u3, u3, u3), 6 O in ±(x, x, z) (x, z, x) (z, x, x); with parameter values u1 = 0.208, u2 = 0.412, u3 = 0.337, x = 0.285 and z = 0.742.
![[alpha]](/logos/entities/alpha_rmgif.gif) = 42° 35' ± 2',
= 42° 35' ± 2', ![[rho]](/logos/entities/rho_rmgif.gif) = 5.25 g.cm-3; Sr3(PO4)2: a = 7.280 ± 0.002 kX,
= 5.25 g.cm-3; Sr3(PO4)2: a = 7.280 ± 0.002 kX, ![[alpha]](/logos/entities/alpha_rmgif.gif) = 43° 21' ± 2', 4.53 g.cm-3. The space group is R
= 43° 21' ± 2', 4.53 g.cm-3. The space group is R
 journal menu
journal menu











