Abstract
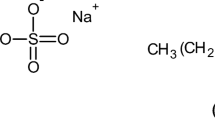
The modification of the triple helical structure in aqueous gelatin solutions by changing pH and adding alkyl sulphates at 298 K and after rechilling the solution to 283 K was investigated by CD-measurement. At 298 K the triple helical content at the IEP of the gelatin has its maximum value. It is only weakly affected by adding sodium dodecyl sulphate (SDDS) at concentrations <10−4 M/dm3. The unfolding of the triple helix affected by pH and SDDS is reversible by rechilling the solution. The triple helical content of gelatin solutions decreases at SDDS concentrations higher than 10−4 M/dm3. In all cases the decrease of the amount of triple helical structure is connected with an increase of the cis-configuration in single chains and leads to chain reversals. At sufficiently high SDDS concentrationsβ-sheets are formed. These changes are thermally irreversible. Sodium decyl sulphate (SDS) has a more minor influence than SDDS except in the range of the c.m.c. of SDS. At sufficiently high SDS concentrations,β-turns appear.
Similar content being viewed by others
References
Küntzel A, Le Nénaon JC (1961) Leder 12:9
Pankhurst KGA, Smith RCM (1944) Trans Faraday Soc 40:565
Ramachandran GN, Kartha G (1954) Nature 174:269
Ramachandran GN, Kartha G (1955) Nature 176:593
Ramachandran GN, Kartha G (1955) Proc Indian Acad Sci 42A:215
Ramachandran GN, Chandrasekharan R (1968) Biopolymers 6:1649
Miller MH, Scheraga HA (1976) J Polymer Sci Symp 54:171
von Hippel PH (1967) In: Ramachandran GN (ed) Treatise on Collagen. Academic Press, New York
Madison V, Schellman J (1972) Biopolymers 11:1041
Ohsaka M, Izumi J, Imamura A (1984) Int J Biol Macromol 6:234
Harrington WF, Rao NV (1970) Biochemistry 9:3714, 3723
Harrington WF, Karr G (1970) Biochemistry 9:3725
Hauschka PV, Harrington WF (1970) Biochemistry 9:3734
Okuyama K, Arnott S, Takayanagi M, Kakudo M (1981) J Mol Biol 152:427
James DW, Rintoul L (1982) Aust J Chem 35:1157
Hoeve CAJ, Lue PC (1982) Biopolymers 21:1661
Lüscher-Mattli M, Ruegg M (1982) Biopolymers 21:403
Gonzales E, Hamabata A, Rojkind M (1984) Collagen Rel Res 4:339
James TC (1980) Teorijay fotograficeskogo processa. Leningrad, Chimija, p 56
Macsuga DD (1972) Biopolymers 11:2521
Gardi A, Nischmann HS, Rieder K (1973) Chimia 27:116
Nishio T, Hayashi R (1985) Agric Biol Chem 49:1675
Mandel R, Holzwarth G (1973) Biopolymers 12:655
Caldwell JW, Applequist J (1984) Biopolymers 23:1891
Sutch K, Noda H (1974) Biopolymers 13:2391
Jennes OD, Sprecher C, Curtis W, Johnson WC (1976) Biopolymers 15:513
Pysh ES (1974) Biopolymers 13:1563
Ronish W, Krimm S (1974) Biopolymers 13:1635
Madison V, Schellman J (1970) Biopolymers 9:65
Applequist J (1981) Biopolymers 20:2311
Wetzel R, Buder E, Hermel H, Hüttner A (1987) Colloid Polym Sci 265:1036
Punkhurst KGA (1949) Disc Faraday Soc 6:52
Tamaki K, Tamamushi B (1955) Bull Chem Soc Jpn 28:555
Knox WJ, Wright JF (1965) J Colloid Sci 20:177
Tavernier BH (1983) J Colloid Interface Sci 93:419
Wüstneck R, Zastrow L, Kretzschmar G (1985) Kolloid Z, USSR 47:462
Wüstneck R, Hermel H, Kretzschmar G (1984) Colloid Polym Sci 262:827
Wüstneck R, Zastrow L, Kretzschmar G (1987) Kolloid Z, USSR 49:10
Wüstneck R, Wüstneck NP, Hermel H, Zastrow L (1987) Kolloid Z, USSR 49:244
Hermel H (1981) Signal AM 9:55
Eastoe JE, Leach AA (1977) In: Ward AG, Courts A (eds) Food Sci and Technology of Gelatin. Academic Press, London New York San Francisco, p 78
Wüstneck R, Fruhner H (1981) Colloid Polym Sci 259:1228
Tamburro AM, Guantieri V (1984) Biopolymers 23:617
Madison V, Schellman J (1972) Biopolymers 11:1041
Brahms S, Brahms J (1980) J Mol Biol 138:149
Wetzel R, Becker M, Behlke J, Billwitz H, Böhm S, Ebert B, Hamann H, Krumbiegel J, Lassmann G (1980) Eur J Biochem 104:469
Tschesche H (1982) In: Hoppe W, Lohmann W, Markl H, Ziegler H (ed) Biophysik. Springer Verlag, Berlin, p 35
Brandts JF, Halvorson HR, Brennan M (1975) Biochemistry 14:4953
Steinberg IZ, Harrington WF, Berger A, Selas M, Katchalski EJ (1960) J Am Chem Soc 82:5263
Pčelin VA, Kulman RA (1961) Vysokomolek Soed 3:768
Izmailova VN, Rehbinder PA (1974) Strukturoobrazovanie v belkovych sistemach. Moskva, Nauka, p 179
Steinhardt J, Reynolds JA (1969) Multiple Equilibria in Proteins. Academic Press, New York
Burkhard RK, Stolzenberg GE (1972) Biochemistry 11:1672
Seno M, Noritomi H, Kuroyanagi Y, Iwamoto K, Ebert G (1984) Colloid Polym Sci 262:727
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wüstneck, R., Wetzel, R., Buder, E. et al. The modification of the triple helical structure of gelatin in aqueous solution I. The influence of anionic surfactants, pH-value, and temperature. Colloid & Polymer Sci 266, 1061–1067 (1988). https://doi.org/10.1007/BF01428818
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01428818