Summary
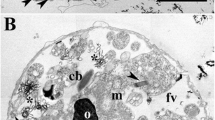
Bundles of decorated tubules found in the sieve elements ofNymphaea have been studied with the transmission electron microscope. Comparatively straight tubules (100 nm in diameter) arise from the endoplasmic reticulum during early stages of sieveelement development and subsequently associate into bundles of up to 100 tubules that parallel the longitudinal cell axis. From the start of their formation the tubules are structurally distinct from other ER profiles due to their dense decoration with particles. High magnifications reveal an orderly array of the particles (about 24 surround a 100 nm tubule) and suggest a modification of their membrane so that it is no longer dissolvable into a regular three-layered structure. Later during sieve-element ontogeny the decorated tubules get invaginated by smooth ER membranes, thereby squeezing out the intratubular (extracytoplasmic) space. As a result a double mantle is formed that surrounds a plasmatic cylinder. Decorated 100 nm tubules with inner membranes are present in enucleate mature sieve elements ofNymphaea alba andN. tuberosa. Considerably larger tubules (about 200 nm in diameter) were found inN. Candida andN. tetragona and occasionally also inNuphar and Barclaya, two other genera from the same family. The decoration of the tubules and their subsequent invagination by smooth membranes are discussed with respect to the controlled autolysis of sieve elements.
Similar content being viewed by others
References
Allen RD (1995) Membrane tabulation and proton pumps. Protoplasma 189: 1–8
Behnke H-D (1968) Zum Aufbau gitterartiger Membranstrukturen im Siebelementplasma vonDioscorea. Protoplasma 66: 287–310
— (1973) Strukturänderungen des Endoplasmatischen Reticulums und Auftreten von Proteinfilamenten während der Siebröhrendifferenzierung beiSmilax excelsa. Protoplasma 77: 279–289
— (1989a) Sieve-element plastids, phloem-proteins and the evolution of flowering plants. IV. Hamamelidae. In: Blackmore S, Crane PR (eds) Systematics, evolution and fossil history of the Hamamelidae, vol 1. Clarendon Press, Oxford, pp 105–128 (Systematics Association special volume 40A)
— (1989b) Structure of the phloem. In: Baker DA, Milburn JA (eds) Transport of photoassimilates. Longman, Harlow, pp 79–137
Dannenbaum C, Schul R (1991) Die Entwicklung der Pollentetraden und Pollinien bei der Asclepiadaceae. Schweizerbart' sehe Verlagsbuchhandlung, Stuttgart (Bibliotheca Botanica 34, issue 141)
Eleftheriou E (1990) Monocotyledons. In: Behnke H-D, Sjolund RD (eds) Sieve elements: comparative structure, induction and development. Springer, Berlin Heidelberg New York Tokyo, pp 139–159
Esau K, Hoefert LL (1980) Endoplasmic reticulum and its relation to microtubules in sieve elements of sugarbeet and spinach. J Ultrastruct Res 71: 249–257
Evert RF (1990) Dicotyledons. In: Behnke H-D, Sjolund RD (eds) Sieve elements: comparative structure, induction and development. Springer, Berlin Heidelberg New York Tokyo, pp 103–137
Karnovsky MJ (1965) A formaldehyde-glutaraldehyde fixation of high osmolality for use in electron microscopy. J Cell Biol 27: 137A-138A
Oparka KJ, Johnson RPC, Bowen ID (1981) Sites of acid phosphatase in the differentiating root protophloem ofNymphoides peltata (S. G. Gmel.) O. Kuntze. Support for the role of stacked ER in sieve-element autolysis. Plant Cell Environ 4: 27–35
Parthasarathy MV (1974a) Ultrastructure of phloem in palms. II. Structural changes, and fate of the organelles in differentiating sieve elements. Protoplasma 79: 93–125
— (1974b) Ultrastructure of phloem in palms. III. Mature phloem. Protoplasma 79: 265–315
Schnepf E, Deichgräber G (1984) Electron microscopic studies of nectaries of someEuphorbia species. In: Rauh W (ed) Anatomisch-biochemische Untersuchungen an Euphorbien, Teil 1. Akademie der Wissenschaften und der Literatur zu Mainz, Mainz, pp 55–93 (Tropische und subtropische Pflanzenwelt, vol 45)
Sjolund RD (1990a) Sieve elements in plant tissue cultures: development, freeze-fracture, and isolation. In: Behnke H-D, Sjolund RD (eds) Sieve elements: comparative structure, induction and development. Springer, Berlin Heidelberg New York Tokyo, pp 179–198
— (1990b) Calcium in phloem sieve element membranes. Curr Top Plant Biochem Physiol 9: 101–118
—, Shih CY (1983) Freeze-fracture analysis of phloem structure in plant tissue cultures. I. The sieve element reticulum. J Ultrastruct Res 82: 111–121
Steer MW, Newcomb EH (1969) Observations on tubules derived from the endoplasmic reticulum in leaf glands ofPhaseolus vulgaris. Protoplasma 67: 33–50
Thorsch J, Esau K (1981a) Changes in the endoplasmic reticulum during differentiation of a sieve element inGossypium hirsutum. J Ultrastruct Res 74: 183–194
— — (1981b) Nuclear degeneration and the association of endoplasmic reticulum with the nuclear envelope and microtubules in maturing sieve elements ofGossypium hirsutum, J Ultrastruct Res 74: 195–204
Wooding FBP (1967) Endoplasmic reticulum aggregates of ordered structure. Planta 76: 205–208
Author information
Authors and Affiliations
Additional information
Dedicated to Prof. Dr. Dr. h.c. Eberhard Schnepf on the occasion of his retirement
Rights and permissions
About this article
Cite this article
Behnke, H.D. Endoplasmic reticulum derived decorated tubules in the sieve elements ofNymphaea . Protoplasma 193, 213–221 (1996). https://doi.org/10.1007/BF01276647
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01276647