Summary
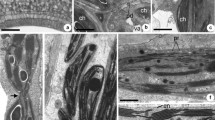
The intracellular localization of partially O-methylated flavonol glucosides was studied inChrysosplenium americanum leaves using caffeine as stabilizing and visualizing reagent. Electron microscopic observations and chromatographic data indicated that intracellular flavonoids were found to accumulate mainly within the walls of epidermal and mesophyll cells. Various membrane profiles and associated vesicles appeared to be involved in the packaging and channelling of the electron-dense material towards the cell wall. There was no evidence to suggest the participation of chloroplasts in these processes. The significance of flavonoid accumulation in cell walls was discussed in relation to the nature of these compounds and their possible ecophysiological role in this plant.
Similar content being viewed by others
Abbreviations
- ER:
-
endoplasmic reticulum
- HPLC:
-
high-performance liquid chromatography
- TLC:
-
thin-layer chromatography
References
Bajaj KL, De Luca V, Khouri H, Ibrahim RK (1983) Purification and properties of flavonol-ring B glucosyltransferase fromChrysosplenium americanum. Plant Physiol 72: 891–896
Bal AK, Savory DR (1980) Characterization of polyphenolcontaining dense cells. Experientia 36: 1292–1294
Baur PS, Walkinshaw CH (1974) Fine structure of tannin accumulations in callus cultures ofPinus elliotti (slash pine). Can J Bot 52: 615–619
Brisson JD, Robb J, Peterson RL (1976) Phenolic localization by ferric chloride and other iron compounds. Proc Microsc Soc Can 3: 174–175
—,Peterson RL, Robb J, Rauser WE, Ellis BE (1977) Correlated phenolic histochemistry using light, transmission and scanning electron microscopy, with examples taken from pathological problems. Scan Electron Microsc 2: 667–676
Brisson L, Vacha WEK, Ibrahim RK (1986) Localization of partially methylated flavonol glucosides inChrysosplenium americanum. II. Immunofluorescence. Plant Sci 44: 175–181
Charriere-Ladreix Y (1977) La secretion flavonique chezPopulus nigra: signification fonctionelle des remaniements ultrastructuraux glandulaires. Physiol Vég 15: 619–640
— (1979) Intracellular distribution of flavonoids in glandular cells. In:Luckner M, Schreiber K (eds) Regulation of secondary product and plant hormone metabolism, FEBS vol 55. Pergamon Press, New York, pp 101–109
Collins FW, De Luca V, Ibrahim RK, Voirin B, Jay M (1981) Polymethylated flavonols ofChrysosplenium americanum: Identification and enzymatic synthesis. Z Naturforsch 36 c: 730–736
De Luca V, Ibrahim RK (1982) Characterization of three distinct flavonol O-methyltransferases fromChrysosplenium americanum. Phytochemistry 21: 1537–1540
— — (1985a) Enzymatic synthesis of polymethylated flavonols inChrysosplenium americanum. I. Partial purification and some properties of S-adenosyl-L-methionine: flavonol 3-, 6-, 7-, and 4′-O-methyltransferases. Arch Biochem Biophys 238: 596–605
— — (1985b) Enzymatic synthesis of polymethylated flavonols inChrysosplenium americanum. II. Substrate interaction and product inhibition studies of flavonol 3-, 6-, and 4′-O-methyltransferases. Arch Biochem Biophys 238: 606–618
Harborne JB (1975) Flavonoid sulfates: A new class of sulphur compounds in higher plants. Phytochemistry 14: 1147–1155
Jensen A (1962) Botanical histochemistry. Freeman, San Francisco
Johnson JE (1982) Your epoxy embedding procedure is the second most important factor in obtaining beautiful electron micrographs of biological specimens. Transmission Scan Electron Microsc J 1982: 10–11
Kartusch B (1975) Gleichzeitiger Nachweis von phenolarmen und phenolreichen Phasen in den Blattepidermiszellen vonSalix alba L. Protoplasma 86: 371–379
Khouri H, Ibrahim RK (1984) Kinetic mechanism of a flavonolring B O-glucosyltransferase fromChrysosplenium americanum. Eur J Biochem 142: 559–564
Martyn RD, Samuelson DA, Freeman TE (1983) Phenol-storing cells in water hyacinth leaves. J Aquat Plant Manage 21: 49–53
McClure JW (1979) The physiology of phenolic compounds in plants. Rec Adv Phytochem 12: 525–556
Mueller WC, Greenwood AD (1978) The ultrastructure of phenolic-storing cells fixed with caffeine. J Exp Bot 29: 757–764
Paul DC, Goff ChW (1973) Comparative effects of caffeine, its analogues and calcium deficiency on cytokinesis. Exp Cell Res 78: 399–413
Roper W, Roper S (1977) Centripetal wall formation in roots ofVicia faba after caffeine treatment. Protoplasma 93: 89–100
Schmid PS, Bartscherer HC, Feucht W (1984) Ultrastructural localization of polyphenols in the sieve tubes ofPrunus avium by ferric chloride. Sci Hortic 22: 105–111
Stafford HA (1981) Compartmentation in natural product biosynthesis by multienzyme complexes. In:Conn EE (ed) The biochemistry of plants, vol 7. Academic Press, New York, pp 117–137
Vaughn KC, Downs BD, Wilson KG (1980) Ultrastructural and cytochemical studies of “air blisters” inPilea cadierei. Ann Bot 46: 221–224
Wagner G (1982) Compartmentation in plant cells: the role of the vacuole. Rec Adv Phytochem 15: 1–45
Wollenweber E (1984) The systematic implication of flavonoids secreted by plants. In:Rodriguez E, Healy P, Metha I (eds) Biology and chemistry of plant trichomes. Plenum Press, New York, pp 53–69
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Charest, P.M., Brisson, L. & Ibrahim, R.K. Ultrastructural features of flavonoid accumulation in leaf cells ofChrysosplenium americanum . Protoplasma 134, 95–101 (1986). https://doi.org/10.1007/BF01275707
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01275707