Abstract
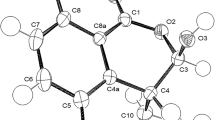
Starting from a chiral substituted benzaldehyde diastereomeric arylmethylidenepyrrolinones were accessible. The geometrical structures of the corresponding isomers were investigated by lanthanide ion induced shift technique and UV spectroscopy. By equilibration studies, D-NMR measurements and kinetic data a complete picture of the various ground state enthalpy differences as well as the activation energies of the transition paths was gained.
The results are in accord with the barriers determined for two simple model derivatives belonging to the class of atropisomeric sterically hindered styrenes.
Similar content being viewed by others
Literatur
Siehe z. B.H. O. Kalinowski undH. Kessler, in: Topics in Stereochemistry (N. L. Allinger undE. L. Eliel, Hrsg.) Bd.7, S. 295. New York: Interscience. 1973.
10. Mitt. in dieser Reihe:R. Adams undJ. W. Mecorney, J. Amer. Chem. Soc.67, 798 (1945).
Z. B.C. S. C. Yang undR. S. H. Liu, Tetrahedron Lett.1973, 4811.
I. O. Sutherland, Ann. Reports on NMR Spectr.4, 71, Academic Press (1971).
J. A. Hirsch, in: Topics in Stereochemistry (N. L. Allinger undE. L. Eliel, Hrsg.), Bd.1, S. 199, New York: Interscience. 1967.
Vgl.5 bzw.H. Günther, NMR-Spektroskopie. Stuttgart: Thieme. 1973.
K. R. Crook undE. W. Wyn-Jones, Trans. Faraday Soc.67, 660 (1971).
QCPE-Programm Nr. 136.
H. Falk, K. Grubmayr undO. Hofer, Mh. Chem.106, 301 (1975);H. Falk, K. Grubmayr, U. Herzig undO. Hofer, Tetrahedron Lett.1975, 559.
H. Falk, S. Gergely undO. Hofer, Mh. Chem.105, 1004 (1974).
H. Falk, K. Grubmayr, O. Hofer undF. Neufingerl, Mh. Chem.106, 991 (1975).
M. R. Willcott III, R. E. Lenkinski undR. E. Davis, J. Amer. Chem. Soc.94, 1742 (1972); Herrn Prof.Davis danken wir für die Überlassung eines PDIGM-Programmdecks sehr herzlich.
H. Falk undJ. M. Ribó, Mh. Chem.107, 307 (1976).
C. W. Haigh undR. B. Mallion, Org. Magn. Res.4, 203 (1972).
J. E. Bloor undN. B. Brearley, Canad. J. Chem.43, 1761 (1965).
Vgl. z. B.H. Kessler undA. Rieker, Ann. Chem.708, 57 (1967);H. Kessler, Ber.103, 973 (1970).
E. B. Barnett undF. G. Sanders, J. Chem. Soc.1933, 434.
A. Rieche, H. Gross undE. Höft, Ber.93, 88 (1960).
H. Plieninger, H. Bauer undA. R. Katrizky, Ann. Chem.654, 165 (1962).
J. H. Atkinson, R. S. Atkinson undA. W. Johnson, J. Chem. Soc.1964, 5999.
Author information
Authors and Affiliations
Additional information
Herrn Prof. Dr.H. Nowotny mit den besten Wünschen zum 65. Geburtstag gewidmet.
Mit 2 Abbildungen
13. Mitt.:H. Falk, K. Grubmayr, O. Hofer, F. Neufingerl undJ. M. Ribó, Mh. Chem.107, 831 (1976).
Rights and permissions
About this article
Cite this article
Falk, H., Hofer, O. & Leodolter, A. Beiträge zur Chemie der Pyrrolpigmente, 14. Mitt. Interkonversionsprozesse bei Arylmethylidenpyrrolinonen; ein Spezialfall atropisomerer, sterisch gehinderter Styrole. Monatshefte für Chemie 107, 907–919 (1976). https://doi.org/10.1007/BF00904477
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00904477