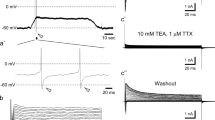
Abstract
The changes in ionic permeability induced by the application of α-latrotoxin to NG108-15 neuroblastoma x glioma cells were examined using the nystatin perforated-patch technique for whole-cell recording. Complex single channel activity appeared in the plasmalemmas after delays that ranged from 1–20 min in Krebs' solution. The conductance of a channel fluctuated among at least three broad, approximately equispaced bands, the maximum conductance being about 300 pS, and the reversal potential approximately 0 mV. The channels were permeable to Na+, K+, Ca2+ and Mg2+, poorly permeable to glucosamineH+ and Cl-, and were blocked by La3+. The channels stayed fully open in Ca2+-free solutions with 4 mm Mg2+, in solutions with no divalent cations and in solutions with 2 mm Ca2+ and 96 mm Mg2+. They opened infrequently if both internal and external Cl- were replaced by glutamate-. If α-latrotoxin opened similar channels in nerve terminals, the flux of ions through them could account for the massive release of neurotransmitter induced by the toxin.
Similar content being viewed by others
References
Ceccarelli, B., Hurlbut, W.P. 1980. Ca2+-dependent recycling of synaptic vesicles at the frog neuromuscular junction. J. Cell Biol. 87:297–303
Ceccarelli, B., Hurlbut, W.P., Iezzi, N. 1988. Effect of α-latrotoxin on the frog neuromuscular junction at low temperature. J. Physiol. 402:195–217
Clark, A.W., Hurlbut, W.P., Mauro, A. 1972. Changes in fine structure of the neuromuscular junction of the frog caused by black widow spider venom. J. Cell Biol. 52:1–14
Clark, A.W., Mauro, A., Longenecker, H.E., Jr., Hurlbut, W.P. 1970. Effects of black widow spider venom on the frog neuromuscular junction: effects on the fine structure. Nature 225:703–705
Delaney, K., Tank, D.W., Zucker, R.S. 1991. Presynaptic calcium and serotonin-mediated enhancement of transmitter release at crayfish neuromuscular junction. J. Neurosci. 11:2631–2643
Del Castillo, J., Pumplin, D.W. 1975. Discrete and discontinuous action of brown widow spider venom on the presynaptic nerve terminals of frog muscle. J. Physiol. 252:491–508
Falke, L.C., Gillis, K.D., Pessel, D.M., Misler, S. 1989. ‘Perforated patch recording’ allows long-term monitoring of metabolite-induced electrical activity and voltage-dependent Ca2+ currents in pancreatic islet B cells. FEBS Lett. 251:167–172
Fenwick, E.M., Marty, A., Neher, E. 1982. Sodium and calcium channels in bovine chromaffin cells. J. Physiol. 331:599–635
Fesce, R., Segal, J.R., Ceccarelli, B., Hurlbut, W.P. 1986. Effects of black widow spider venom and Ca2+ on the quantal secretion at the frog neuromuscular junction. J. Gen. Physiol. 88:737–752
Finkelstein, A., Rubin, L.L., Tzeng, M.-C. 1976. Black widow spider venom: effect of purified toxin on lipid bilayer membranes. Science 193:1009–1011
Frontali, N., Ceccarelli, B., Gorio, A., Mauro, A., Siekevitz, P., Tzeng, M.-C., Hurlbut, W.P. 1976. Purification from black widow spider venom of a factor causing the depletion of synaptic vesicles at the neuromuscular junction. J. Cell Biol. 68:462–479
Grasso, A., Alema, S., Rufini, S., Senni, M.I. 1980. Black widow spider toxin-induced calcium fluxes and transmitter release in a neurosecretory cell line. Nature 283:774–776
Heuser, J.E., Miledi, R. 1971. Effect of lanthanum ions on function and structure of frog neuromuscular junction. Proc. R. Soc. London 179:247–260
Horn, R., Marty, A. 1988. Muscarinic activation of ionic currents measured by a new whole-cell recording method. J. Gen. Physiol. 92:145–159
Larkin, J.M., Brown, M.S., Goldstein, J.L., Anderson, R.G.W. 1983. Depletion of intracellular potassium arrests coated pit formation and receptor-mediated endocytosis in fibroblasts. Cell 33:273–285
Longenecker, H.E., Hurlbut, W.P., Mauro, A., Clark, A.W. 1970. Effects of black widow spider venom on the frog neuromuscular junction: effects on endplate potential, miniature endplate potential and nerve terminal spike. Nature 225:701–703
Mallart, A., Haimann, C. 1985. Differential effects of α-latrotoxin on mouse nerve endings and fibers. Muscle Nerve 8:151–157
Matteoli, M., Haimann, C., Torri Tarelli, F., Polak, J.M., Ceccarelli, B., De Camilli, P. 1988. Differential effect of α-latrotoxin on exocytosis from small synaptic vesicles and large dense-core vesicles containing calcitonin-gene-related peptide at the frog neuromuscular junction. Proc. Natl. Acad. Sci. USA 85:7366–7370
Meldolesi, I. 1982. Studies on α-latrotoxin receptors in rat brain synaptosomes: correlation between toxin binding and stimulation of transmitter release. J. Neurochem. 38:1559–1569
Meldolesi, J., Huttner, W.B., Tsien, R.Y., Pozzan, T. 1984. Free cytoplasmic Ca++ and neurotransmitter release. Studies on PC12 cells and synaptosomes exposed to α-latrotoxin. Proc. Natl. Acad. Sci. USA 81:620–624
Meldolesi, J., Madeddu, L., Torda, M., Gatti, G., Niutta, A. 1983. The effect of α-latrotoxin on the neurosecretory PC12 cell line: studies on toxin binding and stimulation of neurotransmitter release. Neuroscience 10:997–1009
Misler, S., Falke, L.C. 1987. Dependence on multivalent cations of quantal release of transmitter induced by black widow spider venom. Amer. J. Physiol. 253:C469-C476
Misler, S., Hurlbut, W.P. 1979. Action of black widow spider venom on quantal release of acetylcholine at the frog neuromuscular junction: dependence upon external Mg2+. Proc. Nat. Acad. Sci. USA 76:991–995
Nicholls, D.G., Rugolo, M., Scott, I.G., Meldolesi, J. 1982. α-Latrotoxin of black widow spider venom depolarizes the plasma membrane, induces massive calcium influx and stimulates transmitter release in guinea pig brain synaptosomes. Proc. Nat. Acad. Sci. USA 79:7924–7928
Petrenko, A.G. 1993. α-Latrotoxin receptor. Implications in nerve terminal function. FEBS Lett. 325:81–85
Petrenko, A.G., Perin, M.S., Daveltov, B.A., Ushkaryov, Y.A., Geppert, M., Südhof, T.C. 1991. Binding of synaptotagmin to the αlatrotoxin receptor implicates both in synaptic vesicle exocytosis. Nature 353:65–68
Rosenthal, L., Meldolesi, J. 1989. α-Latrotoxin and related toxins. Pharmacol. Ther. 42:115–134
Rosenthal, L., Zacchetti, D., Madeddu, L., Meldolesi, J. 1990. Mode of action of α-latrotoxin: role of divalent cations in Ca2+-dependent and Ca2+-independent effects mediated by the toxin. Mol. Pharmacol. 38:917–923
Scheer, H.W. 1989. Interactions between α-latrotoxin and trivalent cations in rat striatal synaptosomal preparations. J. Neurochem. 52:1590–1597
Scheer, H., Meldolesi, J. 1985. Purification of the putative α-latrotoxin receptor from bovine synaptosomal membranes in an active binding form. EMBO J. 4:323–327
Scheer, H., Prestipino, G., Meldolesi, J. 1986. Reconstruction of the purified α-latrotoxin receptor in liposomes and planar lipid membranes. Clues to the mechanism of toxin action. EMBO J. 5:2643–2648
Segal, J.R., Ceccarelli, B., Fesce, R., Hurlbut, W.P. 1985. Miniature endplate potential frequency and amplitude determined by an extension of Campbell's theorem. Biophys. J. 47:183–202
Smith, J.E., Clark, A.W., Kuster, T.A. 1977. Suppression by elevated calcium of black widow spider venom activity at frog neuromuscular junctions. J. Neurocytol. 6:519–539
Tzeng, M.-C., Siekevitz, P. 1979a. The binding interaction between α-latrotoxin from black widow spider venom and a dog cerebral cortex synaptosomal membrane preparation. J. Neurochem. 33:263–274
Tzeng, M.-C., Siekevitz, P. 1979b. Action of α-latrotoxin from black widow spider venom on a cerebral cortex preparation: release of neurotransmitters, depletion of synaptic vesicles and binding to membrane. In: Advances in Cytopharmacology 3. B. Ceccarelli, and F. Clementi, editors, pp. 117–127. Raven, New York
Ushkaryov, Y.A., Petrenko, A.G., Geppert, M., Südhof, T.C. 1992. Neurexins: synaptic cell surface proteins related to the α-latrotoxin receptor and laminin. Science 257:50–56
Valtorta, F., Madeddu, L., Meldolesi, J., Ceccarelli, B. 1984. Specific localization of the α-latrotoxin receptor in the nerve terminal plasma membrane. J. Cell Biol. 99:124–132
Wanke, E., Ferroni, A., Gattanini, P., Meldolesi, J. 1986. α-Latrotoxin of the black widow spider venom opens a small, non-closing cation channel. Biochem. Biophys. Res. Commun. 134:320–325
Author information
Authors and Affiliations
Additional information
We thank Prof. J. Meldolesi for inviting W.P.H. to the Ceccarelli Center and for supplying the toxin, Prof. F. Clementi for finding financial support for W.P.H., Dr. R. Fesce for the stimulating discussions, Drs. H.-Q. Han and P. Greengard for providing NG108-15 cells and Mrs. Helen Hurlbut for typing the first draft of the manuscript into the computer. W.P.H. is particularly indebted to Prof. S. Misler of Washington University in St. Louis. The pilot experiments for this project were done there, and channels were observed when a crude homogenate of venom glands from American black widow spiders was applied to cultured neuroblastoma cells or to rat chromaffin cells in primary culture.
This work was supported by grants from FIDIA SpA, the Associazione per la Promozione delle Ricerche Neurologiche, the CNR Progetto Strategico Prodotti Biotecnologici (C.H.: CT 90.00005.74.115.24959) and Telethon (F.V.).
Rights and permissions
About this article
Cite this article
Hurlbut, W.P., Chieregatti, E., Valtorta, F. et al. α-Latrotoxin channels in neuroblastoma cells. J. Membarin Biol. 138, 91–102 (1994). https://doi.org/10.1007/BF00211072
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00211072