Abstract
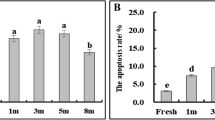
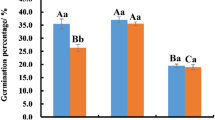
Declining viability of pollen during storage at 24° C in atmospheres of 40% relative humidity (RH) and 75% RH was studied, with special emphasis on lipid changes. Pollens of Papaver rhoeas and Narcissus poeticus, characterized by a high linolenic acid content, were compared with Typha latifolia pollen which has a low linolenic acid content. The rationale behind this was to answer the question of whether lipid peroxidation is involved in the rapid viability loss and reduced membrane integrity of, in particular, the unsaturated-lipid pollen types. Viability and membrane integrity degraded more rapidly at 75% RH than at 40% RH. All pollen species showed deesterification of acyl chains of lipids but no detectable peroxidation at both RH levels. Considerable amounts of lipid-soluble antioxidants were detected that did not degrade during storage. Free fatty acids and lysophospholipids were formed during storage, the effects of which on membranes are discussed. These degradation products were very prominent in the short-lived Papaver pollen. The loss of viability does coincide with phospholipid deesterification. A significant decrease of the phospholipid content occurred at 75% RH, but not at 40% RH. Based on compositional analyses of phospholipids and newly formed free fatty acids, it was concluded that the deesterification of acyl chains from the lipids occurred at random. We suggest that, due to the low water content of the pollen, free radicals rather than unspecific acyl hydrolases are involved in the deesterification process.
Similar content being viewed by others
References
Benson EE (1990) Free radical damage in stored plant germplasm. International Board for Plant Genetic Resources, Rome, pp 20–35
Böttcher CJF, Van Gent CM, Pries C (1961) A rapid and sensitive sub-micro phosphorus determination. Anal Chim Acta 24:203–208
Buchvarov P, Gantchev TS (1984) Influence of accelerated and natural aging on free radical levels in soybean seeds. Physiol Plant 60:53–56
Chan HW-S (1987) Autoxidation of unsaturated lipids. Academic Press, London, pp 17–50
Crowe JH, McKersie BD, Crowe LM (1989) Effects of free fatty acids and transition temperature on the stability of dry liposomes. Biochim Biophys Acta 979:7–10
Faragher JD, Wachtel E, Mayak S (1987) Changes in the physical state of membrane lipids during senescence of rose petals. Plant Physiol 83:1037–1042
Galliard T (1980) Degradation of acyl lipids: hydrolytic and oxidative enzymes.-In: Stumpf PK, Conn EE (eds) The biochemistry of plants, 4, Academic Press, New York, pp 131–161
Hoekstra FA (1986) Water content in relation to stress in pollen. In: Leopold AC (ed) Membranes, metabolism and dry organisms. Comstock, Ithaca, pp 102–122
Hoekstra FA (1992) Stress effects on the male gametophyte. In: Cresti M, Tiezzi A (eds) Sexual Plant Reproduction. Springer Berlin Heidelberg New York, pp 193–201
Hoekstra FA, Bruinsma J (1975) Respiration and vitality of binucleate and trinucleate pollen. Physiol Plant 34:221–225
Hoekstra FA, Van Roekel GC (1985) Correlation of membrane properties to loss of pollen vigor during dry storage. Plant Physiol [Suppl] 77:669
Hoekstra FA, Van Roekel GC (1988) Desiccation tolerance of Papaver dubium L. pollen during its development in the anther. Plant Physiol 88:626–632
Hoekstra FA, Crowe LM, Crowe JH (1989) Differential desiccation tolerance of corn and Pennisetum pollen linked to their sucrose content. Plant Cell Environ 12:83–91
Hoekstra FA, Crowe JH, Crowe LM (1991) Effect of sucrose on phase behaviour of membranes in intact pollen of Typha latifolia L, as measured with Fourier transform infrared spectroscopy. Plant Physiol 97:1073–1079
Hoekstra FA, Crowe JH, Crowe LM (1992) Germination and ion leakage are linked with phase transitions of membrane lipids during imbibition of Typha latifolia pollen. Physiol Plant 84:29–34
Iwanami Y, Nakamura N (1972) Storage in an organic solvent as a means for preserving viability of pollen grains. Stain Technol 47:137–139
McKersie BD, Thompson JE (1978) Phase behaviour of chloroplast and microsomal membranes during leaf senescence. Plant Physiol 61:639–643
McKersie BD, Senaratna T, Walker MA, Kendall EJ, Hetherington PR (1988) Deterioration of membranes during aging in plants: evidence for free radical mediation. In: Noodén LD, Leopold AC (eds) Senescence and aging in plants, Academic Press, New York, pp 441–464
McKersie BD, Hoekstra FA, Krieg LC (1990) Differences in the susceptibility of plant membrane lipids to peroxidation. Biochim Biophys Acta 1030:119–126
Nakayama Y, Saio K, Kito M (1981) Decomposition of phospholipids in soybeans during storage. Cereal Chem 58:260–264
Niehaus WJ Jr (1978) A proposed role of superoxide anion as a biological nucleophile in the deesterification of phospholipids. Bioorg Chem 7:77–84
Pfundt M (1910) Der Einfluss der Luftfeuchtigkeit auf die Lebensdauer des Blütenstaubes. Jahrb Wiss Bot 47:1–40
Priestley DA, Leopold AC (1979) Absence of lipid oxidation during accelerated aging of soybean seeds. Plant Physiol 63:726–729
Priestley DA, Leopold AC (1983) Lipid changes during natural aging of soybean seeds. Physiol Plant 59:476–470
Priestley DA, Werner BG, Leopold AC, McBride MB (1985) Organic free radical levels in seeds and pollen: the effects of hydration and aging. Physiol Plant 64:88–94
Pukacka S, Kuiper JC (1988) Phospholipid composition and fatty acid peroxidation during ageing of Acer platanoides seeds. Physiol Plant 72:89–93
Rosenberg P (1990) Phospholipases. In: Stier WT, Mebs D (eds) Handbook of toxinology. Dekker New York, pp 76–277
Schaich KM (1980) Free radical initiation in proteins and amino acids by ionizing and ultraviolet radiation and lipid oxidation. III. Free radical transfer from oxidizing lipids. CRC Critical Rev Food Sci Nutr 13:189–244
Senaratna T, McKersie BD, Stinson RH (1984) Association between membrane phase properties and dehydration injury in soybean axes. Plant Physiol 76:759–762
Senaratna T, Gusse JF, McKersie BD (1988) Age-induced changes in cellular membranes of imbibed soybean seed axes. Physiol Plant 73:85–91
Shivanna KR, Linskens HF, Cresti M (1991) Response of tobacco pollen to high humidity and heat stress: viability and germinability in vitro and in vivo. Sex Plant Reprod 4:104–109
Van Bilsen DGJL, Hoekstra FA (1993) Decreased membrane integrity in aging Typha latifolia L. pollen: accumulation of lysolipids and free fatty acids. Plant Physiol 101:675–682
Van Bilsen DGJL, Hoekstra FA, Crowe LM, Crowe JH (1994) Altered phase behavior in membranes of aging dry pollen may cause imbibitional leakage. Plant Physiol 104
Wilson DO, McDonald MB (1986) The lipid peroxidation model of seed aging. Seed Sci Technol 14:269–300
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Van Bilsen, D.G.J.L., van Roekel, T. & Hoekstra, F.A. Declining viability and lipid degradation during pollen storage. Sexual Plant Reprod 7, 303–310 (1994). https://doi.org/10.1007/BF00227714
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00227714