Abstract
The alkaline serum phosphatase has been studied by starch gel electrophoresis in 746 healthy individuals, 62 patients with duodenal ulcer, 95 patients with stomach ulcer, and 65 patients with stomach cancer (Fig. 1, Table 1). The previously reported quantitative variations of the B-component of alkaline phosphatase, which is presumably identical with the intestinal component, has been confirmed. The variations of the B-component are associated with the ABO blood group system, the Lewis blood group system, and the ABH secretor status; they are also influenced by diet.
In a sample of 637 healthy, nonfasting persons the B-component was positively correlated to the O, B and Le(a-/b+) blood groups and to ABH secretion. The B-component was negatively associated with the A1 and Le(a+/b-) blood groups and the ABH non-secretor status (Table 2 and 3).
In a sample of 109 fasting healthy individuals the B-component was less frequent and also less intense if present. Nevertheless, the same correlations to the ABO and Lewis blood groups and the ABH secretor status are found as in the non-fasting control (Table 5 and 6).
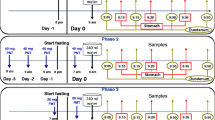
In three persons the increase of the B-component of alkaline phosphatase in serum was followed after intake of a fatty meal (Table 7, Fig. 2 and 3).
In patients with duodenal ulcer, stomach ulcer and stomach cancer the B-component is less frequent and less intense than in the control group of fasting healthy individuals. This deviation is related to differences in the diet or to disturbances of the gastrointestinal functions in these patients. There was no clear indication of a connection between the B-component of alkaline phosphatase and the ABO blood groups association with these gastrointestinal diseases (Table 8, 9, and 10).
Zusammenfassung
Mit Hilfe der Stärkegelelektrophorese wurden die phänotypischen Variationen der alkalischen Serumphosphatase bei 746 Gesunden, 62 Patienten mit Ulcus duodeni, 95 Patienten mit Ulcus ventriculi und 65 Patienten mit Magencarcinom untersucht. Die Beziehungen der quantitativen Variationen der langsamer wandernden B-Komponente zum ABO-Blutgruppensystem, zum Lewis-Blutgruppensystem, zum ABH-Ausscheidersystem und ihre Abhängigkeit von der Nahrungszufuhr werden bestätigt.
Bei der Untersuchung von 637 nicht nüchternen Gesunden wurde eine positive Korrelation der B-Bande zu den Blutgruppen O, B und Le(a-/b+) und den Ausscheidern von ABH-Substanzen gefunden. Eine negative Korrelation der B-Bande fand sich zu den Blutgruppen A1, Le(a+/b-) und den Nichtausscheidern von ABH-Substanzen.
Bei einem Vergleichskollektiv von 109 nüchternen Gesunden wurde im Prinzip wieder die gleiche Korrelation gefunden. Die Häufigkeit und auch die Intensität der B-Bande war jedoch insgesamt geringer als bei den nicht nüchternen Gesunden.
Nach Nahrungsaufnahme kommt es zu einem Anstieg der Intensität der B-Komponente der alkalischen Phosphatase im Serum, wie in einem Diätversuch bei drei Personen gezeigt werden konnte.
Bei Patienten mit Ulcus duodeni, Ulcus ventriculi und Carcinoma ventriculi war sowohl die Häufigkeit als auch die Intensität der B-Bande geringer als bei Gesunden. Diese Abweichung wird auf eine Änderung der Ernährungsweise oder eine Störung im Bereich des Gastrointestinaltraktes zurückgeführt. Für einen Zusammenhang zwischen der B-Komponente der alkalischen Serumphosphatase und der bekannten ABO-Blutgruppenassoziation der drei untersuchten gastrointestinalen Erkrankungen ergab sich kein eindeutiger Hinweis.
Similar content being viewed by others
Literatur
Aird, I., H. H. Bentall, and J. A. F. Roberts: A relationship between cancer of stomach and the ABO blood groups. Brit. med. J. 1953/I, 799–801.
Aird, I., H. H. Bentall, J. A. Mehigan, and J. A. F. Roberts: The blood groups in relation to peptic ulceration and carcinoma of colon, rectum, breast, and bronchus. Brit. med. J. 1954/II, 315–321.
Arfors, K.-E., L. Beckman, and L.-G. Lundin: Genetic variations of human serum phosphatases. Acta genet. (Basel) 13, 89–94 (1963).
Bamford, K. F., H. Harris, J. E. Luffman, E. B. Robson, and T. E. Cleghorn: Serum-alkaline-phosphatase and the ABO blood groups. Lancet 1965/I, 530–531.
Beckman, L.: Association between human serum alkaline phosphatases and blood groups. Acta genet. (Basel) 14, 286–297 (1964).
—: Human alkaline phosphatases and the factors controlling their appearance in serum. Acta genet. (Basel) 16, 305–312 (1966).
Blomstrand, R., and W. Werner: Alkaline phosphate activity in human thoracic duct lymph. Acta chir. scand. 129, 177–191 (1965).
Boyer, S. H.: Alkaline phosphatase in human sera and placentae. Science 134, 1002–1004 (1961).
—: Human organ alkaline phosphatase: Discrimination by several means including starch gel electrophoresis of antienzyme-enzyme supernatant fluids. Ann. N.Y. Acad. Sci. 103, 938–951 (1963).
Chiandussi, L., S. F. Greene, and S. Sherlock: Serum alkaline phosphatase fractions in hepato-biliary and bone diseases. Clin. Sci. 22, 425–434 (1962).
Clarke, C. A.: Blood groups and disease. Progr. med. Genet. 1, 81–119 (1961).
Clarke, C. A. W. K. Cowan, J. W. Edwards, A. W. Howel Evans, R. B. McConnell, J. C. Woodrow, and P. M. Sheppard: The relationship of the ABO blood groups to duodenal and gastric ulceration. Brit. med. J. 1955/II, 643–646.
Cunningham, V. R., and J. G. Rimer: Isoenzymes of alkaline phosphatase of human serum. Biochem. J. 89, 50–51 (1963).
Fishman, W. H., S. Green, and N. I. Inglis: Organ-specific behavior exhibited by rat intestine and liver alkaline phosphatase. Biochim. biophys. Acta (Amst.) 62, 363–375 (1962).
—: L-Phenylalanine: an organ specific, stereospecific inhibitor of human intestinal alkaline phosphatase. Nature (Lond.) 198, 685–686 (1963).
Gahne, B.: Genetic variation of phosphatase in cattle serum. Nature (Lond.) 199, 305–306 (1963).
Gould, B. S.: Studies on the source of serum phosphatase. The nature of the increased serum phosphatase in rats after fat feeding. Arch. Biochem. 4, 175–181 (1944).
Gutman, A. B.: Serum alkaline phosphatase activity in diseases of the skeletal and hepatobiliary systems. A consideration of the current status. Amer. J. Med. 27, 875–901 (1959).
Hodson, A. W., A. L. Latner, and L. Raine: Isoenzymes of alkaline phosphatase. Clin. chim. Acta 7, 255–261 (1962).
Hoppe, H. H.: Die Häufigkeitsverteilung der Blutgruppensysteme ABO, MN, Rh, P und K in Hamburg. Blut 3, 1–14 (1957).
Kay, H. D.: Plasma phosphatase. I. Method of determination. Some properties of the enzyme. J. biol. Chem. 89, 235–247 (1930).
Keiding, N. R.: The alkaline phosphatase fractions of human lymph. Clin. Sci. 26, 291–297 (1964).
Kreisher, J. H., V. A. Close, and H. W. Fishman: Identification by means of L-Phenyl-alanine inhibition of intestinal alkaline phosphatase components separated by starch gel electrophoresis of serum. Clin. chim. Acta 11, 122–127 (1965).
Krüpe, M.: Blutgruppenspezifische pflanzliche Eiweißkörper (Phytagglutinine). Stuttgart: Enke 1956.
Langman, M. J. S., E. Leuthold, E. B. Robson, J. Harris, J. E. Luffman, and H. Harris: Influence of diet on the “intestinal” component of serum alkaline phosphatase in people of different ABO blood groups and secretor status. Nature (Lond.) 212, 41–43 (1966).
Lawrence, S. H., and P. J. Melnick: Enzymatic activity related to human beta-lipoprotein: Histochemical, immuno-electrophoretic and quantitative studies. Proc. Soc. exp. Biol. (N.Y.) 107, 998–1001 (1961).
—: A species comparison of serum proteins and enzymes by starch gel electrophoresis. Proc. Soc. exp. Biol. (N.Y.) 105, 572–575 (1960).
Moss, D. W., R. H. Eaton, J. K. Smith, and L. G. Withby: Alteration in the electrophoretic mobility of alkaline phosphatases after treatment with Neuraminidase. Biochem. J. 98, 32c-33c (1966).
Poulik, M. D.: Starch gel electrophoresis in a discontinuous system of buffers. Nature (Lond.) 180, 1477–1479 (1957).
Price Evans, D. A.: Confirmation of association between ABO blood groups and salivary ABH secretor phenotypes and electrophoretic patterns of serum alkaline phosphatase. J. med. Genet. 2, 126–127 (1965).
Rendel, J., and C. Stormont: Variants of ovine alkaline serum phosphatases and their association with the R-O blood groups (29056). Proc. Soc. exp. Biol. (N.Y.) 115, 853–856 (1964).
Robinson, I. C., and I. E. Pierce: Differential action of neuraminidase on human serum alkaline phosphatase. Nature (Lond.) 204, 472–473 (1964).
Roberts, J. A. F.: Blood groups and susceptibility to disease: A review. Brit. J. prev. soc. Med. 11, 107–125 (1957).
Schlamowitz, M., and O. Bodansky: Tissue sources of human serum alkaline phosphatase, as determined by immunochemical procedures. J. biol. Chem. 234, 1433–1437 (1959).
Shreffler, D. C.: Genetic studies of blood group-associated variations in a human serum alkaline phosphatase. Amer. J. hum. Genet. 17, 71–86 (1965).
Smithies, O.: Zone electrophoresis in starch gels: Group variations in the serum proteins of normal human adults. Biochem. J. 61, 629–641 (1955).
—: An improved procedure for starch-gel electrophoresis: Further variations in the serum proteins of normal individuals. Biochem. J. 71, 585–587 (1959).
Watanabe, K., and W. H. Fishman: Application of stereospecific inhibitor L-Phenylalanine to the enzymorphology of intestinal alkaline phosphatase. J. Histochem. Cytochem. 12, 252–260 (1964).
Author information
Authors and Affiliations
Additional information
Mit Unterstützung durch die Deutsche Forschungsgemeinschaft, Bad Godesberg.
Rights and permissions
About this article
Cite this article
Fichtner, K., Cleve, H., Krüpe, M. et al. Die blutgruppenassoziierten Elektrophoresetypen der alkalischen Serumphosphatase bei Gesunden und bei Patienten mit gastrointestinalen Erkrankungen. Hum Genet 4, 244–261 (1967). https://doi.org/10.1007/BF00292197
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00292197