Summary
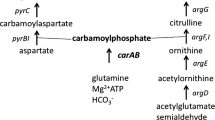
The synthesis of the arginine pathway carbamoylphosphate synthase (CPSase A) of Saccharomyces cerevisiae is subject to two control mechanisms. One mechanism is specific for CPSase A and is exerted by arginine; it probably involves a repressoroperator type of interaction. This “specific” mechanism regulates the expression of gene cpaI coding for the small “glutaminase” subunit of CPSase A but has little influence on the production of the large subunit of the enzyme, a product of gene cpaII. This large component, which alone has no biological significance, accumulates freely under conditions of arginine repression. The second mechanism is general: it controls enzyme synthesis in a number of amino acid biosynthetic pathways in addition to the arginine sequence. Two types of evidence that this “general” mechanism participates in the control of CPSase A synthesis are presented: (1) Derepression upon starvation for any amino acid of which the synthesis is subject to this general control; and (2) repression during growth in amino acid-rich medium. In contrast to the specific mechanism, the “general” mechanism regulates the expression of both the cpaI and cpaII genes.
Similar content being viewed by others
References
Baglioni, C., Campana, T.: α-chain and globin: Intermediates in the synthesis of rabbit hemoglobin. Eur. J. Biochem. 2, 480–492 (1967)
Béchet, J., Grenson, M., Wiame, J. M.: Mutations affecting the repressibility of arginine biosynthetic enzymes in Saccharomyces cerevisiae. Eur. J. Biochem. 12, 31–39 (1970)
Carsiotis, M., Jones, R.F.: Cross-pathway regulation: tryptophanmediated control of histidine and arginine biosynthetic enzymes in Neurospora crassa. J. Bacteriol. 119, 889–892 (1974a)
Carsiotis, M., Jones, R.F., Wesseling, A.C.: Cross-pathway regulation: histidine-mediated control of histidine, tryptophan and arginine biosynthetic enzymes in Neurospora crassa. J. Bacteriol. 119, 893–898 (1974b)
Cybis, J., Davis, R.H.: Organization and control in the arginine biosynthetic pathway of Neurospora. J. Bacteriol. 123, 196–202 (1975)
Delforge, J., Messenguy, F., Wiame, J.M.: The regulation of arginine biosynthesis in Saccharomyces cerevisiae: the specificity of argR mutations and the general control of amino acid biosynthesis. Eur. J. Biochem. 57, 231–239 (1975)
Hilger, F.: Construction and analysis of tetraploid yeast sets for gene dosage studies. J. Gen. Microbiol. 75, 23–31 (1973)
Hilger, F., Culot, M., Minet, M., Piérard, A., Grenson, M., Wiame, J.M.: Studies on the kinetics of the enzyme sequence mediating arginine biosynthesis in Saccharomyces cerevisiae. J. Gen. Microbiol. 75, 33–41 (1973)
Lacroute, F.: Regulation of pyrimidine biosynthesis in Saccharomyces cerevisiae. J. Bacteriol. 95, 824–832 (1968)
Lacroute, F., Piérard, A., Grenson, M., Wiame, J.M.: The biosynthesis of carbamoylphosphate in Saccharomyces cerevisiae. J. Gen. Microbiol. 40, 127–142 (1965)
Lacroute, F., Slonimski, P.P.: Etude physiologique de mutants résistants au 5-fluorouracile chez la levure. C.R. Acad. Sci. (Paris) 258, 2172–2174 (1964)
Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J.: Protein measurements with the Folin phenol reagent. J. Biol. Chem. 193, 265–275 (1951)
Messenguy, F.: Concerted repression of the synthesis of the arginine biosynthetic enzymes by aminoacids: A comparison between the regulatory mechanisms controlling aminoacid biosyntheses in bacteria and in yeast. Mol. Gen. Genet. 169, 85–95 (1979)
Messenguy, F., Cooper, T.G.: Evidence that the specific and “general” control of ornithine carbamoyltransferase production occurs at the level of transcirption in Saccharomyces cerevisiae. J. Bacteriol. 130, 1253–1261 (1977)
Piérard, A., Glansdorff, N., Gigot, D., Crabeel, M., Halleux, P., Thiry, L.: Repression of Escherichia coli carbamoylphosphate synthase: Relationships with enzyme synthesis in the arginine and pyrimidine pathways. J. Bacteriol. 127, 291–301 (1976)
Piérard, A., Grenson, M., Glansdorff, N., Wiame, J.M.: A comparison of the organization of carbamoylphosphate synthesis in Saccharomyces cerevisiae and Escherichia coli, based on genetical and biochemical evidences. In: The enzymes of glutamine metabolism, S. Prusiner, E.R. Stadtman, eds., pp. 483–503. New York: Academic Press 1973
Piérard, A., Schröter, B.: Structure-function relationships in arginine pathway carbamoylphosphate synthase of Saccharomyces cerevisiae. J. Bacteriol. 134, 167–176 (1978)
Ramos, F., Thuriaux, P., Wiame, J.M., Béchet, J.: The participation of ornithine and citrulline in the regulation of arginine metabolism in Saccharomyces cerevisiae. Eur. J. Biochem. 12, 40–47 (1970)
Ramos, F., Wiame, J.M.: Synthesis and activation of asparagine in asparagine auxotrophs of Saccharomyces cerevisiae. Eur. J. Biochem. 94, 409–417 (1979)
Stephens, J.C., Artz, S.W., Ames, B.N.: Guanosine 5′-diphosphate 3′-diphosphate (ppGpp): positive effector for histidine operon transcription and general signal for amino acid deficiency. Proc. Natl. Acad. Sci. U.S.A. 72, 4389–4393 (1975)
Thuriaux, P., Ramos, F., Pierard, A., Grenson, M., Wiame, J.M.: Regulation of the carbamoylphosphate synthetase belonging to the arginine biosynthetic pathway of Saccharomyces cerevisiae. J. Mol. Biol. 67, 277–287 (1972)
Author information
Authors and Affiliations
Additional information
Communicated by W. Gajewski
Rights and permissions
About this article
Cite this article
Piérard, A., Messenguy, F., Feller, A. et al. Dual regulation of the synthesis of the arginine pathway carbamoylphosphate synthase of Saccharomyces cerevisiae by specific and general controls of amino acid biosynthesis. Molec. Gen. Genet. 174, 163–171 (1979). https://doi.org/10.1007/BF00268353
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00268353