Abstract
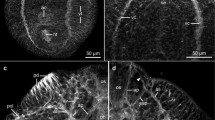
Although the presence of phosphorylcholine (PC) inTrichinella is well established, the structures of the TSL-4 antigens that bear this epitope are unknown. A subset of TSL-4 antigens (TSL-8 antigens) has been reported to be absent from the surface of first-stageT. spiralis larvae. We report experiments with a monoclonal antibody (mAb US2) developed in mice with a relative inability to produce antibodies to PC. In immunoblotting, mAb US2 and anti-PC mAb (BH8) showed apparently identical binding patterns. In addition, we used an immunogold double-labeling technique to study the anatomical distribution of the epitopes recognized by these mAbs; the results obtained indicate close colocalization of epitopes for BH8 and US2 in tissues ofT. spiralis first-stage larvae. On the basis of these results, we suggest that US2 probably binds to allT. spiralis TSL-4 antigens, including TSL-8 antigens. We also clarify some conflicting previous reports on the distribution of PC immunoreactivity in first-stage larvae ofT. spiralis.
Similar content being viewed by others
References
Appleton JA, Bell RG, Homan W, Van Knapen F (1991) Consensus onTrichinella spiralis antigens and antibodies. Parasitol Today 7:190–192
Baltar P, Leiro J, Santamarina MT, Sanmartín ML, Porto MC, Ubeira FM (1991) Specific immunosuppression byTrichinella: fine specifically and effect on lymphocyte function in vivo. Parasitology 102:411–418
Chesebro B, Metzger H (1972) Affinity labeling of a PC binding mouse myeloma protein. Biochemistry 11:766–771
Claflin JL, Hudak S, Maddalena A, Bender T (1985) Antigen-specific anti-phosphocholine antibodies: binding site studies. J Immunol 134:2536–2543
Estévez J, Leiro J, Santamarina MT, Domínguez J, Ubeira FM (1994) Monoclonal antibodies to turbot (Scophthalmus maximus) immunoglobulins: characterization and applicability in immunoassays. Vet Immunol Immunopathol 41:353–366
Faro J, Seoane R, Puentes E, Ubeira FM, Regueiro BJ (1985) Immunoresponses toNeisseria meningitidis epitopes: primary versus secondary antiphosphorylcholine responses. Infect Immun 48:428–432
Feng S, Stein KE (1991) VH gene family expression in mice with the xid defect. J Exp Med 174:45–51
Fletcher TC, White A, Baldo BA (1980) Isolation of a phosphorylcholine-containing component from the turbot tapeworm,B. scorpii (Müller), and its reaction with C-reactive protein. Parasite Immunol 27:102–110
García-Martín LO, Abad M, Sánchez-López JL, Galarza A (1982) Posible agregado multienzimático con actividad glucolítica en glándula digestiva de mejillón,Mytilus galloprovincialis, Lamarck. Rev Esp Fisiol 38:51–58
Homan WL, Derksen ACG, Van Knapen F (1993) Phosphorylcholine onTrichinella spiralis antigens (abstract). Proceedings, 8th International Conference on Trichinellosis, September 7–10, Orvieto, Italy, p 70
Itoh M, Sato S (1991) Comparative studies on biotin-coupled components in adult worms ofParagonimus species. Jpn J Parasitol 40:383–387
Kenny JJ, Guelde G, Claflin JL, Scher I (1981) Altered idiotype response to phosphorylcholine in mice bearing and X-linked immune defect. J Immunol 127:1629–1633
Köhler H, Smyk S, Fung J (1981) Immune response to phosphorylcholine. VIII. The response of CBA/N mice to PC-LPS. J Immunol 126:1790–1793
Lal RB, Ottensen EA (1989) Phosphocholine epitopes on helminth and protozoal parasites and their pesence in the circulation of infected human patients. Trans R Soc Trop Med Hyg 83:652–655
Maizels RM, Selkirk ME (1989) Immunobiology of nematode antigens. In: Eglund PT, Sher A (eds) The biology of parasitism, vol 9. Alan R. Liss, New York, pp 285–308
Maizels RM, Burke J, Denham DA (1987) Phosphorylcholinebearing antigens in filarial nematode parasites: analysis of somatic extracts, in vitro secretions and infection sera fromBrugia malayi andB. pahangi. Parasite Immunol 9:49–66
Moses V (1978) Compartmentation of glycolysis inEscherichia coli. In: Srere PA, Estabrook (eds) Microenvironments and metabolic compartmentation. Academic Press, New York, pp 169–186
Pozio E, La Rosa G, Murrell D, Lichtenfels R (1992) Taxonomic revision of the genusTrichinella. J Parasitol 78:654–659
Romarís E, Baltar P, Estévez JM, Sanmartín ML, Ubeira FM (1994) Estudio de la reactividad cruzada de varios anticuerpos monoclonales anti-T. spiralis. Proceedings, X Reunión Anual de la Asociación de Parasitólogos Españoles, September 23–24, Barcelona, Spain, p 80
Sanmartín ML, Iglesias R, Santamarina MT, Leiro J, Ubeira FM (1991) Anatomical location of phosphorylcholine and other antigens on encystedTrichinella using immunohistochemistry followed by Wheatley's trichrome stain. Parasitol Res 77:301–306
Takahashi Y, Uno T, Mizuno N, Yamada S, Araki T (1989) Ultrastructural localization of the antigenic substances inTrichinella spiralis. Parasitol Res 75:316–324
Takahashi Y, Homan W, Lim PL (1993) Ultrastructural location of the phosphorylcholine-associated antigen inTrichinella spinalis. J Parasitol 79:604–609
Vos T de, Dick TA (1993) The mucosal and systemic response to phosphorylcholine in mice infected withTrichinella spiralis. Exp Parasitol 76:401–411
Ubeira FM, Leiro J, Santamarina MT Villa TG, Sanmartín ML (1987) Immune response toTrichinella epitopes: the antiphosphorylcholine plaque-forming cell response during the biological cycle. Parasitology 94:543–553
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hernández, S., Romarís, F., Acosta, I. et al. Ultrastructural colocalization of phosphorylcholine and a phosphorylcholine-associated epitope in first-stage larvae ofTrichinella spiralis . Parasitol Res 81, 643–650 (1995). https://doi.org/10.1007/BF00931840
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00931840