Summary
Observations on the formation of nuclear membranes and of the changing ultrastructural appearance during mitosis in developing eggs ofLocusta migratoria are reported.
-
1.
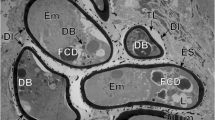
Nuclear membranes are formed while the mitotic chromosomes are still in a condensed state. How exactly the membranes are formed, is not yet understood. Vesicles which occur in the interchromosomal space might participate in the membrane formation but they might just as well appear simultaneously with the membranes.
-
2.
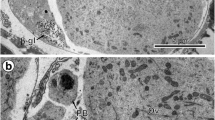
Depending on the developmental stage of the egg, differences in the way in which the interphase nucleus is built up, are found. In early cleavage, karyomeres are formed from single chromosomes which are individually covered by a double membrane. These karyomeres expand by uncoiling their chromosomal mass and subsequently fuse to form one nucleus. In embryonic cells of the germinal rudiment the double envelope is formed only after the chromosomes have clustered and have formed the telophase figure.
-
3.
Nuclear membranes are formed in close contact with the condensed chromosomes. Only after the membranes have been built up, the chromosomes lose their electron density. Chromosomes in early developmental stages (cleavage) uncoil in karyomeres, although the mitotic cycle may only have reached anaphase. In cells from later stages (germinal rudiment), where the chromosomes cluster before membrane formation occurs, the membrane formation as well as the uncoiling are postponed to telophase. This behavior may be related to changes in the duration of the mitotic cycle, indicating progressive differentiation.
-
4.
During the growth of karyomeres, chromosomes lose most of their electron density. Dense cores of 0.2 Μ in diameter, however, persist in the karyomeres of cleavage energides and may prove the existence of a chromosomal axis in certain mitotic stages.
-
5.
Nucleoli are not found in karyomeres during early cleavage stages. Prenucleoli are observed to form at telophase in the later stages of development. More prenucleoli seem to occur in telophase than do nucleoli in interphase nuclei.
Zusammenfassung
Die Formierung der Kernmembran und das wechselnde Erscheinungs-bild der Feinstrukturen von Chromosomen während einiger Phasen des Mitosezyklus wurden an verschiedenen Entwicklungsstadien des Eies vonLocusta migratoria beobachtet.
-
1.
Kernmembranen werden gebildet, solange die Chromosomen noch in kondensiertem Zustand vorliegen. Bisher bleibt ungeklärt, auf welche Weise sie zustande kommen. Die zahlreichen Vesikel, die in der Umgebung der Chromosomen gefunden werden, könnten daran beteiligt sein, könnten aber ebenso gut gleichzeitig mit den Kernmembranen in diesem Bereich auftreten.
-
2.
Die Rekonstruktion des Kerns findet, je nach Entwicklungsstadium, in unterschiedlicher Weise statt. In frühen Furchungsstadien werden, durch Ausbildung von Doppelmembranen um Einzelchromosomen, Karyomeren gebildet. Diese nehmen an Ausdehnung zu, während sich die Chromosomen in ihrem Inneren entspiralisieren, bis schlie\lich die Karyomeren miteinander zum Interphasekern verschmelzen. In Keimanlagenzellen wird dagegen die Doppelmembran erst ausgebildet, nachdem die Chromosomen sich zur Telophasefigur zusammengeschart haben. In diesem einheitlichen Kern entspiralisieren sich die Chromosomen.
-
3.
Erst nach der Ausbildung der Kernmembran verlieren die Chromosomen ihre Elektronendichte. In frühen Entwicklungsstadien erfolgt die Entspiralisation in den Karyomeren, unabhängig davon, da\ sich die Energide erst im Anaphasestadium befindet. In Zellen der Keimanlage wird sowohl die Ausbildung der Kernmembran als auch die Entpiralisation der Chromosomen bis zur Telophase verzögert. Diese Beobachtung mag mit der Verlängerung des Mitosezyklus in Zusammenhang stehen und eine fortgeschrittene Differenzierung andeuten.
-
4.
Während der Ausbildung der Karyomeren verlieren die Chromosomen fast ihre gesamte Elektronendichte. Nur ein Strang von etwa 0,2 Μ Durchmesser, anscheinend spiralförmig in der Längsachse des Karyomers angeordnet, behält seine verdichtete Konsistenz während der Stadien der Karyomerenbildung und -verschmelzung bei. Dieser Strang könnte die Existenz einer häufig geforderten Chromosomenachse beweisen. Möglicherweise liegt die Achse in dieser Form nur in bestimmten Mitosestadien vor.
-
5.
In den Karyomeren der frühen Furchungsstadien sind Nucleoli nicht beobachtet worden. An den noch verdichteten Chromosomen der späteren Entwicklungsstadien fallen dichtere Bezirke auf, die als Praenucleoli gedeutet werden. Allerdings erscheinen davon mehr als zwei, während im Interphasekern nur zwei Nucleoli auftreten.
Similar content being viewed by others
References
Balinsky, B. I.: The role of cortical granules in the formation of the fertilization membrane and the surface membrane of fertilized sea urchin eggs. Symposium on Germ Cells and Development, Pavia 1963, p. 550–563.
Barer, R., S. Joseph, andG. A. Meek: The origin and fate of the nuclear membrane in meiosis. Proc. roy. Soc. B152, 353–366 (1960).
— — —: The origin of the nuclear membrane. Exp. Cell Res.18, 179–182 (1959).
Bernhard, W., andE. de Harven: L'ultrastructure du centriole et d'autres éléments de l'appareil achromatique. Proc. 4th Intern. Conf. Electron Microscopy, vol. 2, p. 217–227. Berlin-Göttingen-Heidelberg: Springer 1960.
Brown, D. D.: The nucleus and synthesis of ribosomal RNA during oogenesis and embryogenesis ofXenopus laevis. Nat. Cancer Inst. Monogr.23, p. 297–309. Ed. W. S. Vincent and O. L. Miller, Jr. Bethesda, Md. 1966.
Buck, R.: Lamellae in the spindle of mitotic cells of Walker 256 Carcinoma. J. biophys. biochem. Cytol.11, 227–236 (1961).
Cooper, K. W.: The nuclear cytology of the grass mite,Pediculopsis graminum (Reut.), with special reference to karyomerokinesis. Chromosoma (Berl.)1, 51–103 (1939).
Das, N. K., andM. Alfert: Detection of a nucleolar component and its behavior during the mitotic cycle. Anat. Rec.134, 548–549 (1959).
Gaulden, M. E.: The nucleolus and mitotic activity. In: The cell nucleus. Ed. J. S. Mitchell. The Faraday Society. London: Butterworths 1960.
Gibbons, J. R.: Chromosomal structure in primary spermatocytes of the locust. Proc. 4th Intern. Conf. Electron Microscopy, vol. 2, p. 238. Berlin-Göttingen-Heidelberg: Springer 1960.
Harris, P.: Electron microscope study of mitosis in sea urchin blastomeres. J. biophys. biochem. Cytol.11, 419–431 (1961).
—, andD. Mazia: The finer structure of the mitotic apparatus. In: Symposia of the Intern. Soc. for Cell Biology, vol. 1, The Interpretation of Ultrastructure. Ed. R. J. C. Harris. New York and London: Academic Press 1962.
Hess, O., andG. F. Meyer: Chromosomal differentiations of the lampbrush type formed by the chromosome inDrosophila hydei andDrosophila neohydei. J. Cell Biol.16, 527–539 (1963).
Ito, S.: The lamellar systems of cytoplasmic membranes in dividing spermatogenic cells ofDrosophila virilis. J. biophys. biochem. Cytol.7, 433–442 (1960).
John, B., andS. A. Henderson: Asynapsis und Polyploidie inSchistocera paranensis. Chromosoma (Berl.)13, 111–147 (1962).
Kunz, W.: Lampenbürstenchromosomen und multiple Nucleolen bei Orthopteren. Chromosoma (Berl.)21, 446–462 (1967).
Lafontaine, J. G., andL. A. Chouinard: A correlated light and electron microscopy study of the nucleolar material during mitosis inVicia faba. J. Cell Biol.17, 167–201 (1963).
Mahowald, A. P.: Fixation problems for electron microscopy ofDrosophila embryos. Dros. Inf. Service36, 130–131 (1962).
Mazia, D.: Mitosis and the physiology of cell division. In: The cell, vol. 3. Ed. J. Brachet, and A. E. Mirsky. New York and London: Academic Press 1961.
Moses, M. J.: The relation between the axial complex of meiotic prophase chromosomes and chromosome pairing in a salamander (Plethodon cinereus). J. biophys. biochem. Cytol.4, 633–638 (1958).
—: Breakdown and reformation of the nuclear envelope at cell division. Proc. 4th Intern. Conf. Electron Microscopy, vol. 2, p. 230–233. Berlin-Göttingen-Heidelberg: Springer 1960.
—, andJ. R. Coleman: Structural patterns and the functional organization of chromosomes. In: The role of chromosomes in development. Ed.: M. Locke. New York and London: Academic Press 1964.
Nebel, B. R., andE. M. Coulon: Enzyme effects on pachytene chromosomes of the male pigeon evaluated with the electron microscope. Chromosoma (Berl.)13, 292–299 (1962).
Nolte, D. J.: The nuclear phenotype of locusts. Chromosoma (Berl.)15, 367–388 (1964).
Porter, K. R.: Problems in the study of nuclear fine structure. Proc. 4th Intern. Conf. Electron Microscopy, vol. 2, p. 186–199. Berlin-Göttingen-Heidelberg: Springer 1960.
—: The ground substance. Observations from electron microscopy. In: The cell, vol. 2, p. 621–677. Ed. J. Brachet, and A. E. Mirsky. New York and London: Academic Press 1962.
—, andR. Machado: Studies on endoplasmatic reticulum IV: Its form and distribution during mitosis in cells of the onion root tip. J. biophys. biochem. Cytol.7, 167–180 (1960).
Reynolds, E. S.: The use of lead citrate at high pH as an electron opaque stain in electron microscopy. J. Cell Biol.17, 208 (1963).
Ris, H.: The structure of meiotic chromosomes in the grasshopper and its bearing on the nature of chromosomes and lamp-brush chromosomes. Biol. Bull.89, 242–257 (1945).
Robbins, E., andN. K. Gonatas: The ultrastructure of a mammalian cell during the mitotio cycle. J. Cell Biol.21, 429–463 (1964).
Ruthmann, A.: The fine structure of the meiotic spindle of the crayfish. J. biophys. biochem. Cytol.5, 177–179 (1959).
Sauer, H. W.: Zeitraffer-Mikrofilm Analyse embryonaler Differenzierungsprozesse vonGryllus domesticus. Z. Morph. ökol. Tiere56, 143–251 (1966).
Schwalm, F. E.: Zell- und Mitosenmuster der normalen und nach Röntgenbestrahlung regulierenden Keimanlage vonGryllus domesticus. Z. Morph. ökol. Tiere55, 915–1023 (1965).
Seidel, F.: Entwicklungsphysiologie der Tiere. Berlin:W. de Gruyter & Co. 1953.
Sotelo, J. R., andR. Wettstein: Fine structure of meiotic chromosomes ofGryllus argentinus. Experientia (Basel)20, 610–612 (1964).
Taylor, J. H.: Chromosome reproduction. Int. Rev. Cytol.13, 39–74 (1962).
Watson, M. L.: The nuclear envelope. Its structure and relation to cytoplasmic membranes. J. biophys. biochem. Cytol.1, 257–270 (1955).
Wettstein, R., andJ. R. Sotelo: Fine structure of meiotic chromosomes. The elementary components of metaphase chromosomes ofGryllus argentinus. J. Ultrastruct. Res.13, 367–381 (1965).
Yasuzumi, G.: Electron microscopy of the nuclear membrane in prophase and telophase in Yoshida sarcoma cells. Z. Zellforsch.50, 110–120 (1959).
Zamboni, L., andL. Mastroianne, Jr: Electron microscopic studies on rabbit ova. II.: The penetrated tubal ovum. J. Ultrastruct. Res.14, 118–132 (1966).
Author information
Authors and Affiliations
Additional information
These studies were mainly carried out during the tenure of an Advanced Research Fellowship from the Anglo American Corporation of South Africa. They were continued under N.S.F. Grant GB 4847 X. I thank the director of the Zoology Department at the University of the Witwatersrand, Prof. Dr. B. I.Balinsky and his staff for their hospitality and assistance during my stay in South Africa. Thanks are also due to the Professors Dr.Bodenstein and Dr.Kretsinger, for reading the manuscript.
Rights and permissions
About this article
Cite this article
Schwalm, F.E. Changes in the formation of nuclear membranes and the ultrastructure of chromosomes during the early development of locust eggs (Locusta migratoria). W. Roux' Archiv f. Entwicklungsmechanik 162, 41–55 (1969). https://doi.org/10.1007/BF00581545
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00581545