Abstract
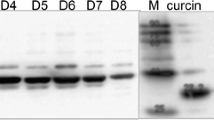
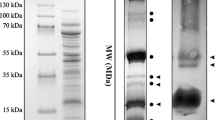
Two new N-glycosidase type-1 ribosome-inactivating proteins (RIPs), denoted petroglaucin 1 and petrograndin, respectively, were isolated from the plantsPetrocoptis glaucifolia (Lag.) Boiss sp.viscosa (Rothm.) Laínz andPetrocoptis grandiflora Rothm. These new RIPs do not share H2N-terminal amino-acid sequence homology with petroglaucin (now denoted as petroglaucin 2), the only other type-1 RIP to be isolated fromP. glaucifolia (Arias et al. (1992) Planta186, 532–540). Petroglaucin 1 shares amino-acid sequence homology with RIPs from Cucurbitaceae while petroglaucin 2 and petrograndin do so with saporins and dianthin 30 (Caryophyllaceae). The new RIPs strongly inhibited protein synthesis at subnanomolar concentrations in rabbit reticulocyte lysates and other eukaryotic cell-free systems, but they were inactive on bacterial ribosomes.
Similar content being viewed by others
Abbreviations
- RIP:
-
ribosome-inactivating protein
References
Arias, F.J., Rojo, M.A., Ferreras, J.M., Iglesias, R., Muñoz, R., Girbés, T. (1992a) Preparation and optimization of a cell-free translation system fromVicia sativa germ lacking ribosome-inactivating protein activity. J. Exp. Bot. 43, 729–737
Arias, F.J., Rojo, M.A., Ferreras, J.M., Iglesias, R., Muñoz, R., Rocher, A., Méndez, E., Barbieri, L., Girbés, T. (1992b) Isolation and partial characterization of a new ribosome-inactivating protein fromPetrocoptis glaucifolia (Lag.) Boiss. Planta186, 532–540
Barbieri, L., Stoppa, C., Bolognesi, A. (1987) Large scale chromatographic purification of ribosome-inactivating proteins. J. Chromatogr. 408, 235–243
Brigotti, M., Rambelli, F., Zamboni, M., Montanaro, L., Sperti, S. (1989) Effect of α-sarcin and ribosome-inactivating proteins on the interaction of elongation factors with ribosomes. Biochem. J. 257, 723–727
Byers, V.S., Levin, A.S., Waites, L.A., Starrett, V.A., Mayer, R.A., Clegg, J.A., Price, M.R., Robins, R.A., Delany, M., Baldwin, R.W. (1990) A phase I/II study of trichosanthin treatment of HIV disease. AIDS4, 1189–1196
Carnicelli, D., Brigotti, M., Montanaro, L., Sperti, S. (1992) Differential requirements of ATP and extra-ribosomal proteins by eight RNA N-glycosidases. Biochem. Biophys. Res. Commun. 182, 579–582
Citores, L., Ferreras, J.M., Iglesias, R., Carbajales, M.L., Arias, F.J., Jiménez, P., Rojo, M.A., Girbés, T. (1993) Molecular mechanism of inhibition of mammalian protein synthesis by some four-chain agglutinis. Proposal of an extended classification of plant ribosomeinactivating proteins (rRNA N-glycosidases). FEBS Lett. 329, 59–62
Collins, E.J., Robertus, J.D., Presti, M.L., Stone, K.L., Williams, K.R., Wu, P., Hwang, K., Piatak, M. (1990) Primary amino acid sequence of α-trichosanthin and molecular models for abrin A-chain and α-trichosanthin. J. Biol. Chem. 256, 8665–8669
Ferreras, J.M., Iglesias, R., Merino, M.J., Girbés, T. (1989) Presence of translational inhibitory activity in partially purified extracts from twoPetrocoptis species. Cell. Mol. Biol. 35, 89–95
Ferreras, J.M., Barbieri, L., Girbés, T., Battelli, M.G., Rojo, M.A., Arias, F.J., Rocher, M.A., Soriano, F., Méndez, E., Stirpe, F. (1993) Distribution and properties of major ribosome-inactivating proteins (28 S rRNA N-glycosidases) of the plantSaponaria officinalis L. (Caryophyllaceae). Biochem. Biophys. Acta. 1216, 31–42
Frankel, A.E., ed. (1988) Immunotoxins. Kluwier Academic Publishers, Boston
Girbés, T., Barbieri, L., Ferreras, J.M., Arias, F.J., Rojo, M.A., Iglesias, R., Alegre, C., Escarmis, C., Stirpe, F (1993a) Effects of ribosomeinactivating proteins onEscherichia coli andAgrobacterium tumefaciens translation systems. J. Bacteriol. 175, 6721–6724
Girbés, T., Citores, L., Ferreras, J.M., Rojo, M.A., Iglesias, I., Muñoz, R., Arias, F.J., Calonge, M., García, J.R., Méndez, E. (1993b) Isolation and partial characterization of nigrin b, a non-toxic novel type 2 ribosome-inactivating protein from the bark of Sambucus nigra L. Plant Mol. Biol. 22, 1181–1186
Girbés, T., Citores, L., Iglesias, R., Ferreras, J.M., Muñoz, R., Rojo, M.A., Arias, F.J., García, J.R., Méndez, E., Calonge, M. (1993c) Ebulin 1, a non-toxic novel type-2 ribosome-inactivating protein fromSambucus ebulus L. leaves. J. Biol. Chem. 268, 18195–18199
Habuka, N., Akiyama, K., Tsuge, H., Miyano, M., Matsumoto, T., Noma, M. (1990) Expression and secretion of Mirabilis antiviral protein in Escherichia coli and its inhibition of in vitro eukaryotic and prokaryotic protein synthesis. J. Biol. Chem. 265, 10988–10992
Hartley, M.R., Legname, G, Osborn, R., Chen, Z., Lord, J.M. (1991) Single-chain ribosome inactivating proteins from plants depurinateEscherichia coli 23 S ribosomal RNA. FEBS Lett. 290, 65–68
Iglesias, R., Arias, F.J., Rojo, M.A., Escarmis, C., Ferreras, J.M., Girbés, T. (1993a) Molecular action of the type 1 ribosome-inactivating protein saporin 5 onVicia sativa ribosomes. FEBS Lett. 325, 291–294
Iglesias, R., Escarmis, C., Alegre, C., Ferreras, J.M., Girbés, T. (1993b) Fusidic acid-dependent ribosomal complexes protectEscherichia coli ribosomes from the action of the type 1 ribosome-inactivating protein crotin 2. FEBS Lett. 318, 189–192
Ippoliti, R., Lendaro, E., Bellelli, A., Brunori, M. (1992) A ribosomal protein is specifically recognized by saporin, a plant toxin which inhibits protein synthesis. FEBS Lett. 298, 145–148
Islam, M.R., Nishida, H., Funatsu, G. (1990) Complete amino acid sequence of luffin-a, a ribosome-inactivating protein from the seeds of sponge gourd (Luffa cylindrica). Agric. Biol. Chem. 54, 2967–2978
Kung, S.S., Kumura, M., Funatsu, G. (1990) The complete amino acid sequence of antiviral protein from the seeds of Pokeweed (Phytolacca americana). Agric. Biol. Chem. 54, 3301–3318
Leah, R., Tommerup, H., Svendsen, I., Mundy, J. (1991) Biochemical and molecular characterization of three barley seed proteins with antifungal properties. J. Biol. Chem. 266, 1564–1573
Lee-Huang, S., Huang, P.L., Kung, H.F., Li, B.Q., Huang, P., Huang, H.L., Chen, H.C. (1991) An anti-human immunodeficiency virus protein fromTrichosanthes kirilowii that is nontoxic to intact cells. Proc. Natl. Acad. Sci. USA88, 6570–6574
Legname, G., Bellosta, P., Gromo, G., Modena, D., Keen, J.N., Roberts, L.M., Lord, J.M. (1991) Nucleotide sequence of cDNA coding for dianthin 30, a ribosome-inactivating protein fromDianthus caryophyllus. Biochem. Biophys. Acta1090, 119–122
Lodge, J.K., Kaniewski, W.K., Turner, N.E. (1993) Broad-spectrum virus resistance in transgenic plants expressing pokeweed antivirial protein. Proc. Natl. Acad. Sci. USA90, 7089–7093
Logemann, J., Jach, G., Tommerup, H., Mundy, J., Schell, J. (1992) Expression of a barley ribosome-inactivating protein leads to increased fungal protection in transgenic tobacco plants. Bio/technology10, 305–308
McGrath, M.S., Hwang, K.M., Caldwell, S.E., Gaston, I., Luk, K, Wu, P., Ng, V.L., Crowe, S., Daniels, J., Marsh, J., Deinhart, T., Lekas, V.P., Vennari, J.C., Yeung, H.W., Lifson, J.D. (1989) GLQ223: an inhibitor of human immunodeficiency virus replication in acutely and chronically infected cells of lymphocyte and mononuclear phagocyte lineage. Proc. Natl. Acad. Sci. USA86, 2844–2848
Merino, M.J., Ferreras, J.M., Muñoz, R., Iglesias, R., Girbés, T. (1990) Plant species containing inhibitors of eukaryotic protein synthesis. J. Exp. Bot. 41, 67–70
Minami, Y., Funatsu, G (1993) The complete aminoacid sequence of momordin-a, a ribosome-inactivating protein from the seeds of bitter gourd (Momordica charantia). Biosci. Biotech. Biochem. 57, 1141–1144
Moazed, D., Robertson, J.M., Noller, H.F. (1988) Interaction of elongation factors EF-G and EF-Tu with a conserved loop in 23 S RNA. Nature334, 362–364
Montecucchi, P.C., Lazzarini, A.M., Barbieri, L., Stirpe, F., Soria, M., Lappi, D. (1989) N-terminal sequence of some ribosome-inactivating proteins. Int. J. Peptide Protein Res. 33, 263–267
Prestle, J., Hornung, E., Schönfelder, M., Mundry, K.W. (1992a) Mechanism and site of action of a ribosome-inactivating protein type 1 fromDianthus barbatus which inactivatesEscherichia coli ribosomes. FEBS Lett. 297, 250–252
Prestle, J., Schönfelder, M., Adam, G., Mundry, K.W. (1992b) Type 1 ribosome-inactivating proteins depurinate plant 25S rRNA without spiecies specificity. Nucleic Acids Res. 20, 3179–3182
Ready, M.P., Brown, D.T., Robertus, J.D. (1986) Extracellular localization of pokeweed antiviral protein. Proc. Natl. Acad. Sci. USA83, 5053–5056
Rojo, M.A., Arias, F.J., Iglesias, R., Ferreras, J.M., Muñoz, R., Girbés, T. (1993) ACucumis sativus cell-free translation system: preparation, optimization and sensitivity to some antibiotics and ribosome-inactivating proteins. Physiol. Plant. 88, 549–556
Stirpe, F., Barbieri, L., Battelli, M.G., Soria, M., Lappi, D.A. (1992) Ribosome-inactivating proteins from plants: present status and future prospects. Bio/technology10, 405–412
Zarling, J.M., Moiran, P.A., Haffar, O., Sias, J., Richman, D.D., Spina, C.A., Myers, D., Kuebelbeck, V., Ledbetter, J.A., Uckun, F.M. (1990) Inhibition of HIV replication by pokeweed antiviral protein targeted to CD4+ cells by monoclonal antibodies. Nature347, 92–95
Author information
Authors and Affiliations
Additional information
This work was supported by grants from CICYT (BIO89-0705 and BIO92-0231) and the Junta de Castilla y León (Spain) to T. Girbés and FIS (92-0858) to E. Méndez. We wish to thank N. Skinner (University of Salamanca, Salamanca, Spain) for correcting the English version of the manuscript. The technical assistance of J. E. Basterrechea is also greatly appreciated.
Rights and permissions
About this article
Cite this article
Arias, F.J., Rojo, M.A., Ferreras, J.M. et al. Isolation and characterization of two new N-glycosidase type-1 ribosome-inactivating proteins, unrelated in amino-acid sequence, from Petrocoptis species. Planta 194, 487–491 (1994). https://doi.org/10.1007/BF00714460
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00714460