Summary
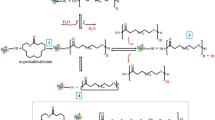
We have previously demonstrated an efficient polyester synthesis of ω-hydroxyacids catalysed by the Candida rugosa lipase in dry hexane (1,2). This method is now extended to the polymerisation of racemic 10-hydroxyundecanoic acid to explore the polymerisation of a secondary alcohol and the ability of the lipase to display stereoselectivity. The resultant polyester (∼1000 Mwt) contained monomer units with an average enantiomeric excess of 60%ee favouring the (S) enantiomer. The residual monomer was recovered with a 33%ee favouring the (R) enantiomer. The origin of this stereoselectivity has been evaluated and it has been shown to arise due to the immediate chiral environment surrounding the esterification event between the acylated enzyme and the secondary alcohol nucleophile. It is not due to recognition of the remote secondary alcohol functional group by the enzyme during initial acylation.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 18 August 1998/Accepted: 24 September 1998
Rights and permissions
About this article
Cite this article
O'Hagan, D., Parker, A. Enzyme mediated polyester synthesis with the lipase from Candida rugosaPreparation of an enantiomerically enriched polymer from an A-B monomer. Polymer Bulletin 41, 519–524 (1998). https://doi.org/10.1007/s002890050396
Issue Date:
DOI: https://doi.org/10.1007/s002890050396