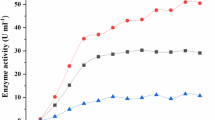
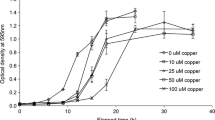
Abstract
Homoveratric acid (HVA) degradation was observed in cultures of Pleurotus eryngii lacking lignin peroxidase (LiP) activity. Extracellular enzymes seemed responsible for this transformation, and the lack of activity after ultrafiltration of the culture liquid suggests that the presence of some low-molecular-size compounds is required. This hypothesis is supported by rapid HVA transformation after addition of the synthetic laccase substrate 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) to the ultrafiltered liquid. HVA transformation by the extracellular enzymes from P. eryngii takes place via Cα-Cβ breakdown and formation of veratryl alcohol, which is further transformed into veratraldehyde. The same major compounds were found during HVA transformation by LiP from Phanerochaete chrysosporium, but this reaction was not stimulated by ABTS. Although the involvement of other enzymes cannot be ruled out, purified laccase from Pleurotus eryngii caused the same HVA transformation pattern in presence of ABTS. Moreover, veratryl alcohol oxidation by P. eryngii laccase was demonstrated in the presence of ABTS. These results suggest that enzymatic systems lacking LiP could be responsible for natural degradation of lignin.
Similar content being viewed by others
References
Archibald FS (1992) A new assay for lignin-type peroxidases employing the dye Azure-B. Appl Environ Microbiol 58:3110–3116
Bourbonnais R, Paice MG (1988) Veratryl alcohol oxidases from the lignin degrading basidiomycete Pleurotus sajor-caju Biochem J 255:445–450
Bourbonnais R, Paice MG (1990) Oxidation of non-phenolic substrates. An expanded role for laccase in lignin biodegradation. FEBS Lett 267:99–102
Bourbonnais R, Paice MG (1992) Demethylation and delignification of Kraft pulp by Trametes versicolor laccase in the presence of 2,2′-azinobis-(3-ethylbenzthiazoline-6-sulphonate). Appl Microbiol Biotechnol 36:823–827
Dodson PJ, Evans CS, Harvey PJ, Palmer JM (1987) Production and properties of an extracellular peroxidase from Coriolus versicolor which catalyses Cα-Cβ cleavage in a lignin model compound. FEMS Microbiol Lett 42:17–22
Guillén F, Martínez AT, Martínez MJ (1990) Production of hydrogen peroxide by aryl-alcohol oxidase from the ligninolytic fungus Pleurotus eryngii. Appl Microbiol Biotechnol 32:465–469
Guillén F, Martínez AT, Martínez MJ (1992) Substrate specificity and properties of the aryl-alcohol oxidase from the ligninolytic fungus Pleurotus eryngii. Eur J Biochem 209:603–611
Gutiérrez A, Caramelo L, Prieto A, Martínez MJ, Martínez AT (1994) Anisaldehyde production and aryl-alcohol oxidase and dehydrogenase activities in ligninolytic fungi from the genus Pleurotus. Appl Environ Microbiol 60 (6), in press
Kamra DN, Zadrăzil F (1986) Influence of gaseous phase, light and substrate pretreatment on fruit-body formation, lignin degradation and in vitro digestibility of wheat straw fermented with Pleurotus spp. Agric Wastes 18:1–17
Kawai S, Umezawa T, Shimada M, Higuchi T, Koide K, Nishida T, Morohoshi N, Haraguchi T (1987) Cα-Cβ cleavage of phenolic β-1 lignin substructure model compound by laccase of Coriolus versicolor. Mokuzai Gakkaishi 33:792–797
Kawai S, Umezawa T, Higuchi T (1989) Oxidation of methoxylated benzyl alcohols by laccase of Coriolus versicolor in the presence of syringaldehyde. Wood Res 76:10–16
Kirk TK, Farrell RL (1987) Enzymatic “combustion”: The microbiol degradation of lignin. Annu Rev Microbiol 41:465–505
Kimura Y, Asada Y, Kuwahara M (1990) Screening of basidiomycetes for lignin peroxidase genes using a DNA probe. Appl Microbiol Biotechnol 32:436–442
Kühn S, Camarero S, Valmaseda M, Almendros G, Martínez MJ, González AE, Martínez AT (1992) Straw biopulping: selective delignification with Pleurotus eryngii. In: Kuwahara M, Shimada M (eds) Biotechnology in the pulb and paper industry. UNI, Tokyo, pp 15–20
Martínez AT, Camarero S, Guillén F, Gutiérrez A, Muñoz C, Varela E, Martínez MJ, Barrasa JM, Ruel K, Pelayo M (1994) Progress in biopulping of non-woody materials: chemical, enzymatic and ultrastructural aspects of wheat-straw delignification with ligninolytic fungi from the genus Pleurotus. FEMS Microbiol Rev 13: in press
Muheim A, Leisola MSA, Schoemaker HE (1990) Aryl-alcohol oxidase and lignin peroxidase from the white-rot fungus Bjerkandera adusta. J Biotechnol 13:159–167
Muheim A, Fiechter A, Harvey PJ, Schoemaker HE (1992) On the mechanism of oxidation of non-phenolic lignin model compounds by the laccase-ABTS couple. Holzforschung 46:121–126
Muñoz C, Martínez AT, Martínez MJ (1993) Induction and partial purification of laccase from Pleurotus eryngii. In: Duarte JC, Ferreira MC, Ander P (eds) Proceedings of the FEMS Symposium on Lignin Biodegradation and Transformation, Lisbon, 17–21 April. Forbitec Editions, Lisbon, pp 173–174
Niku-Paavola M-L, Karhunen E, Salola P, Raunio V (1988) Ligninolytic enzymes of the white-rot fungus Phlebia radiata. Biochem J 254:877–884
Paice MG, Reid OD, Bourbonnais R, Archibald FS, Jurasek L (1993) Manganese peroxidase, produced by Trametes versicolor during pulp bleaching, demethylates and delignifies Kraft pulp. Appl Environ Microbiol 59:260–265
Pérez J, Jeffries TW (1990) Mineralization of 14C-ring-labeled synthetic lignin correlates with the production of lignin perxidase, not of manganese peroxidase or laccase. Appl Environ Microbiol 56:1806–1812
Périé FH, Gold MH (1991) Manganese regulation of manganese peroxidase expression and lignin degradation by the white rot fungus Dichomitus squalens. Appl Environ Microbiol 57:2240–2245
Sannia G, Giardina P, Luna M, Rossi M, Buonocore W (1986)_ Laccase from Pleurotus ostreatus. Biotechnol Lett 8:797–800
Sannia G, Limongi, Cocca E, Buonocore F, Nitt G, Giardina P (1991) Purification and characterization of a veratryl alcohol oxidase enzyme from the lignin degrading basidiomycete Pleurotus ostreatus Biochim Biophys Acta 1073:114–119
Schoemaker HE, Tuor U, Muheim A, Schmidt HWH, Leisola MSA (1991) White-rot degradation of lignin and xenobiotics. In: Betts WB (eds) Biodegradation: natural and synthetic matterials. Springer Berlin, Heidelberg, New York, London pp 157–174
Shimada M, Higuchi T (1991) Microbial, enzymatic and biomimetic degradation of lignin. In: Hon DN-S, Shiraiski N (eds) Wood and cellulosic chemistry. Dekker, New York, pp 557–619
Tien M, Kirk TK (1988) Lignin peroxidase of Phanerochaete chrysosporium. Methods Enzymol 161B:238–248
Valmaseda M, Almendros G, Martínez AT (1990) Substrate-dependent degradation patterns in the decay of wheat straw and beech wood by ligninolytic fungi. Appl Microbiol Biotechnol 33:481–484
Valmaseda M, Almendros G, Martínez AT (1991a) Chemical transformation of wheat straw constituents after solid-state fermentation with selected lignocellulose-degrading fungi. Biomass Bioenergy 1:261–266
Valmaseda M, Martínez MJ, Martínez AT (1991b) Kinetics of wheat straw solid-state fermentation with Trametes versicolor and Pleurotus ostreatus−lignin and polysaccharide alteration and production of related enzymatic activities. Appl Microbiol Biotechnol 35:817–823
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Martínez, M.J., Muñoz, C., Guillen, F. et al. Studies on homoveratric acid transformation by the ligninolytic fungus Pleurotus eryngii . Appl Microbiol Biotechnol 41, 500–504 (1994). https://doi.org/10.1007/BF00178479
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00178479