Abstract
The cycloaddition of ethylene to butadiene has been studied by theab initio LCAO-SCF-MO method of Roothaan using STO-3G and 7s-3p basis sets.
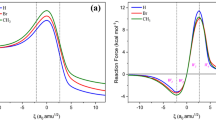
The potential energy hypersurface of the supersystem formed by the reactants has been calculated in order to determine the reaction path. It was found that, during the approach of the partners, the planes of the molecules form an angle around 70 °.
The activated complex has a geometry which prefigures the half-chair conformation of cyclohexene and exhibits no biradical character. Our theoretical results are in reasonable agreement with the corresponding experimental ones.
Similar content being viewed by others
References
McCabe,J.R., Eckert,C.A.: Acc. Chem. Res.7, 251 (1974)
Dewar,M.J.S., Griffin,A.C., Kirschner,S.: J. Am. Chem. Soc., Com. 6225 (1974)
Hehre,W.J., Stewart,R.F., Pople,J.A.: J. Chem. Phys.51, 2657 (1969)
Clementi,E., Clementi,M., Davis,D.R.: J. Chem. Phys.46, 4725 (1967)
GAUSSIAN-70 from Pople's group adapted by Claude,L., Peeters,D., Sana,M.
Boys,S.J.: Rev. Mod. Phys.32, 296 (1960)
Magnasco,V., Perico,A.: J. Chem. Phys.47, 971 (1967)
Chiang,J.F., Bauer,S.H.: J. Am. Chem. Soc.91, 1898 (1969)
Bucourt,R., Hainaut,D.: Bull. Soc. Chim. France12, 4562 (1967)
Mulliken,R.S.: J. Chem. Phys.23, 1833 (1955)
Leroy,G., Sana,M.: Theoret. Chim. Acta (Berl.)33, 329 (1974)
Sana,M.: Ph. D. Thesis, Louvain (1975)
Rowley,D., Steiner,H.: Disc. Farad. Soc.10, 198 (1951)
Smith,S.R., Gordon,A.S.: J. Phys. Chem.65, 1124 (1961)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Burke, L.A., Leroy, G. & Sana, M. Theoretical study of the Diels-Alder reaction. Theoret. Chim. Acta 40, 313–321 (1975). https://doi.org/10.1007/BF00668337
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00668337