Abstract
Purpose. To develop and validate internally an in vitro-in vivo correlation (IVIVC) for a hydrophilic matrix extended release metoprolol tablet.
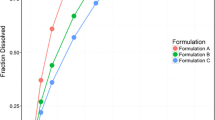
Methods. In vitro dissolution of the metoprolol tablets was examined using the following methods: Apparatus II, pH 1.2 & 6.8 at 50 rpm and Apparatus I, pH 6.8, at 100 and 150 rpm. Seven healthy subjects received three metoprolol formulations (100 mg): slow, moderate, fast releasing and an oral solution (50 mg). Serial blood samples were collected over 48 hours and analyzed by a validated HPLC assay using fluorescence detection. The f 2 metric (similarity factor) was used to analyze the dissolution data. Correlation models were developed using pooled fraction dissolved (FRD) and fraction absorbed (FRA) data from various combinations of the formulations. Predicted metoprolol concentrations were obtained by convolution of the in vivo dissolution rates. Prediction errors were estimated for Cmax and AUC to determine the validity of the correlation.
Results. Apparatus I operated at 150 rpm, and pH of 6.8 was found to be the most discriminating dissolution method. There was a significant linear relationship between FRD and FRA when using either two or three of the formulations. An average percent prediction error for Cmax and AUC for all formulations of less than 10% was found for all IVIVC models.
Conclusions. The relatively low prediction errors for Cmax and AUC observed strongly suggest that the metoprolol IVIVC models are valid. The average percent prediction error of less than 10% indicates that the correlation is predictive and allows the associated dissolution data to be used as a surrogate for bioavailability studies.
Similar content being viewed by others
REFERENCES
In Vitro In Vivo Evaluation of Dosage Forms., United States Pharmacopeia XXIII chapter 1088, United States Pharmacopeial Convention, Incorporated, Rockville, Maryland, pp. 1927–1929
J. P. Skelly, G. L. Amidon, W. H. Barr, L. Z. Benet, J. E. Carter, J. R. Robinson, V. P. Shah, and A. Yacobi. Report of the workshop on in-vitro and in-vivo testing and correlation for oral controlled/modified-release dosage forms. J. Pharm. Sci. 79: 849–854 (1990).
Guidance for the Industry: Extended Release Solid Oral Dosage Forms: Development, Evaluation and Application of In Vitro/In Vivo Correlations. U. S. Department of Health, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), September, 26, 1997.
J. H. Silas, S. Freestone, M. S. Lennard, and L. E. Ramsay. Comparison of two slow-release formulations of metoprolol with conventional metoprolol and atenolol in hypertensive patients. Br. J. Clin. Pharmacol. 20:387–391 (1985).
A. Sandberg, B. Abrahamsson, A. Svenheden, B. Olofsson, and R. Bergstrand. Steady-state bioavailability and day-to-day variability of a multiple unit (CR/ZOK) and a single-unit (OROS) delivery system of metoprolol after once-daily dosing. Pharm. Res. 10:28–34 (1993).
A. Sandberg, G. Ragnarsson, U. E. Jonsson, and J. Sjörgren. Design of a new multiple-unit controlled-release formulation of metoprolol-CR. Eur. J. Clin. Pharmacol. 33:Supp S3–S7 (1988).
G. Amidon, H. Lennernas, V. P. Shah, and J. R. Crison. A theoretical basis for a biopharmaceutical drug classification: The correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm. Res. 12:413–420 (1995).
G. S. Rekhi, N. D. Eddington, M. J. Fossler, P. Schwartz, P., L. J. Lesko, and L. L. Augsburger. Evaluation of In Vitro Release Rate and In Vivo Absorption characteristics of Four Metoprolol Tartrate Immediate Release Tablet Formulations. Pharmaceutical Dev. Tech. 29:11–24 (1997).
R. V. Nellore, G. S. Rekhi, A. S. Hussain, L. G. Tillman, and L. L. Augsburger. Development of Metoprolol Tartrate Extended-Release Matrix Tablet formulations for Regulatory Consideration. J. Controlled Release (in press).
Modified Release Solid Oral Dosage Form Guidance: Scale-up and Postapproval Changes: Chemistry, Manufacturing and Controls, In Vitro Dissolution Testing and In Vivo Bioequivalence Documentation. U. S. Department of Health, Food and Drug Administration, Center for Drug Evaluation and Research (CDER), September, 26, 1997.
M. Hildebrand, W. Seifert, and A. Reichenberger. Determination of dextromethorphan metabolizer phenotype in healthy volunteers. Euro. J. Clin. Pharmacol. 1989:315–318 (1989).
B. Mistry, J. Leslie, and N. D. Eddington. A sensitive assay of metoprolol and its major metabolite, α-hydroxy metoprolol in human plasma and determination of dextromethorphan and its metabolite in urine with high performance liquid chromatography and fluorometric detection. J. Pharmaceut. Biomed. Anal. (in press).
J. W. Moore and H. H. Flanner. Mathematical comparison of curves with an emphasis on dissolution profiles. Pharm. Tech. 6:64–74 (1996).
N. H. G. Holford and L. B. Sheiner. Understanding the dose-effect relationship: clinical application of pharmacokinetic-pharmacodynamic models. Clin. Pharmacokin. 6:429–453 (1981).
L. B. Sheiner and S. Beal. Some suggestions for measuring predictive performance. J. Pharmacokinet. Biopharm. 9:503–512 (1981).
J. P. Skelly, G. A. Van Buskirk, H. M. Arbit, G. L. Amidon, L. L. Augsburger, W. H. Barr, S. Berge, J. Clevenger, S. Dighe, M. Fawzi, D. Fox, M. A. Gonzalez, V. A. Gray, C. Hoiberg, L. J. Lesson, L. Lesko, H. Malinowski, P. R. Nixon, D. M. Pearce, G. Peck, S. Porter, J. Robinson, D. R. Savello, P. Schwartz, J. B. Schwartz, V. P. Shah, R. Shangraw, F. Theeuwes, and T. Wheatley. Workshop II Report. Scaleup of oral extended-release dosage forms Pharm. Res. 10:1800–1805 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Eddington, N.D., Marroum, P., Uppoor, R. et al. Development and Internal Validation of an In Vitro-in Vivo Correlation for a Hydrophilic Metoprolol Tartrate Extended Release Tablet Formulation. Pharm Res 15, 466–473 (1998). https://doi.org/10.1023/A:1011988601696
Issue Date:
DOI: https://doi.org/10.1023/A:1011988601696