Abstract
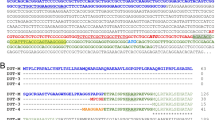
During muscle, heart, and brain neonatal maturation, the capacity to utilize glucose in energy metabolism is directly related to the extent of accumulation of the 6-phosphofructo-1-kinase (PFK) M-type subunit. Neonatal development of other organs, such as liver and kidney, which are not characterized by large increases in the capacity to use glucose do not exhibit large increases in the M-type subunit protein. The presence of the M-type subunit in a PFK isozyme pool fosters a higher affinity utilization of carbohydrate and increased responsiveness to the levels of regulatory metabolites. To better appreciate this phenomenon, which is vital for normal development, the different isoforms of the M-type subunit mRNA's and alteration of their levels during maturation have been examined. Further, the potential promoter regions, i.e., the regions upstream from the sites of initiation of transcription, which are involved in expression of the different M-type subunit mRNA isoforms have been isolated, sequenced, and examined for possible transcription factor interaction sites. Using cDNA libraries produced from adult rat brain or skeletal muscle RNA, two primary forms of rat M-type subunit cDNA's were detected. Although the translated regions of these mRNA's were essentially identical, the 5′-untranslated region (5′-UTR) exhibited different lengths (90 or 59 bp) and sequences. Each M-type subunit cDNA had 10 common nucleotides immediately upstream from the initiator ATG, and the remaining 5′-UTR's had insignificant identity. A genomic fragment which interacted with probes complimentary to the sequences of the 5′-UTR of each M-type subunit mRNA isoform was isolated and sequenced by primer walking. It was discovered that the 5′-UTR of one of the mRNA's (proximal mRNA) was located immediately upstream from exon I and was apparently transcribed without splicing. Subsequently, the initial bp in the sequence of the other mRNA isoform (distal mRNA) was located 4010 bp upstream from the ATG in exon 1. Employing Reverse Transcription-Polymerase Chain Reaction using total RNA and scanning densitometry, the relative levels of the proximal and distal mRNA's during neonatal maturation of brain, heart, and muscle were measured. In these tissues, both forms of M-type subunit mRNA's were present, and during maturation tissue-specific differences were noted.
Similar content being viewed by others
References
Dunaway GA: A review of animal phosphofructokinase isozymes with an emphasis on their physiological role. Mole Cell Biochem 52: 75–96, 1983
Foe LG, Kemp RG: Isozyme composition and phosphorylation of brain phosphofructokinase. Arch Biochem Biophys 228: 503–511, 1984
Dunaway GA, Kasten TP: Nature of the rat brain 6-phosphofructo-1-kinase isozymes. J Biol Chem 260: 4180–4185, 1985
Dunaway GA, Kasten TP: Characterization of the rat heart 6-phosphofructo-1-kinase isozymes. J Mol Cell Cardiol 17: 947–957, 1985
Thrasher JR, Cooper MD, Dunaway GA: Developmental changes in the heart and muscle phosphofructokinase isozymes. J Biol Chem 256: 7844–7848, 1981
Vora S, Seaman C, Durham S, Piomelli S: Isozymes of human phosphofructokinase: Identification and subunit structural characterization of a new system. Proc Natl Acad Sci USA 77: 62–66, 1980
Kahn A, Meienhofer MC, Cottreau D, LaGrange JL, Dreyfus JC: Phosphofructokinase (PFK) isozymes in man I. studies in adult human tissues. Hum Genet 48: 93–108, 1979
Meienhofer MC, LaGrange JL, Cottreau, D, Lenoir G, Dreyfus JC, Kahn A: Phosphofructokinase in human cells. Blood 54: 389–400, 1979
Dunaway GA, Kasten TP, Nickols GA, Chesky JA: Regulation of skeletal muscle 6-phosphofructo-1-kinase during aging and development. Mech Age Dev 36: 13–23, 1986
Dunaway GA, Kasten TP: Physiological relevance of the changing subunit composition and regulatory properties of the 6-phosphofructo-1-kinase isozyme pools during heart and muscle development. Mol Cell. Biochem 87: 71–77, 1989
Dunaway G, Kasten T: Physiological implications of the alteration of 6-phosphofructo-1-kinase isozyme pools during brain development and aging. Brain Res 456: 310–316, 1988
Kasten TP, Mhaskar Y, Dunaway GA: Regulation of brain 6-phosphofructo-1-kinase: Effects of aging, fructose-2,6-bisphosphate, and regional subunit distribution. Mol Cell Biochem 120: 61–66, 1993
Close RI: Dynamic properties of mammalian skeletal muscle. Physiol Rev 52: 129–197, 1972
Dunaway GA, Kasten TP, Kolm P: Alteration of 6-phosphofructo-1-kinase isozyme pools during heart development and aging. J Biol Chem 261: 17170–17173, 1987
Mourek J: Oxidative metabolism of nervous tissue during ontogeny in rat. In: W.A. Himwich (ed). Developmental Neurobiology. C.C. Thomas, Springfield, IL, 1970, pp 370–392
Ontell M: The growth and metabolism of developing muscle. In: C.T. Jones (ed). The Biochemical Development of the Fetus and Neonate. Elsevier Biomedical Press, NY, 1982, pp 213–247
Lee C-P, Kao M.C, French B, Putney S, Chang S: The rabbit muscle phosphofructokinase gene. J Biol Chem 262: 4195–4199, 1987
Yamasaki T, Nakajima H, Kono N, Hotta K, Yamada K, Imai E, Kuwajima M, Noguchi T, Tanaka T, Tarui S: Structure of the entire human muscle phosphofructokinase-encoding gene: A two-promoter system. Gene 104: 277–282, 1991
Vaisanen PA, Reddy GR, Sharma PC, Kohani R, Johnson JL, Raney AK, Babior BM, McLachian A: Cloning and characterization of the human muscle phosphofructokinase gene. DNA Cell Biol 11: 461–470, 1992
Valdez BC, Chen Z, Sosa MG, Younathan EZ, Chang S: Human 6-phosphofructo-1-kinase gene has an additional intron upstream of start codon. Gene 76: 167–169, 1989
Nakajima H, Yamasaki T, Noguchi T, Tanaka T, Kono N, Tarui S: Evidence for alternate splicing and possible alternative promoters in the human muscle phosphofructokinase gene at the 5′ untranslated region. Biochem Biophys Res Commun 166: 637–641, 1990
Li J, Chen Z, Lu L, Byrnes M, Chang S: Sequence diversity in the 5′ untranslated region of rabbit muscle phosphofructokinase mRNA. Biochem Biophys Res Commun 170: 1056–1060, 1990
Sanger F, Nicklen S, Coulson AR: DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci USA 74: 5463–5467, 1977
Mhaskar Y, Dunaway GA: Alteration of 6-phosphofructo-1-kinase subunit protein, synthesis, and mRNA during neonatal brain development. Dev Brain Res 85: 54–57, 1995
Mhaskar Y, Dunaway GA: Alteration of 6-phosphofructo-1-kinase subunit protein, synthesis, and mRNA during rat neonatal development. Mech Age Dev 86: 161–172, 1996
Nakajima H, Noguchi T, Hamaguchi T, Tomita K, Hanafusa T, Kono N, Tanaka T, Kuwajima M, Matsuzawa Y: Expression of mouse phosphofructokinase-M gene alternate transcripts: Evidence for the conserved two promoter system. Biochem J 303: 449–453, 1994
Dunaway GA, Kasten TP, Crabtree S, Mhaskar Y: Age-related changes in subunit composition and regulation of hepatic 6-phosphofructo-1-kinase. Biochem J 266: 823–827, 1988
Nakajima H, Kono N, Yamasaki T, Hotta K, Kuwajima M, Noguchi T, Tanaka T, Tarui S: Tissue specificity in expression and alternative RNA splicing of human phosphofructokinase-M and-L genes. Biochem Biophys Res Commun 173: 1317–1321, 1990
Quandt K, Frech K, Karas H, Wingender E, Werner T: MatInd and MatInspector — new and versatile tools for detection of consensus matches to nucleotide sequence data. Nucl Acid Res 23: 4878–4884, 1995
Lefrancois-Martinez A-M, Martinez A, Antoine B, Raymondjean M, Kahn A: Upstream stimulatory factor proteins are major components of the glucose response complex of the l-type subunit pyruvate kinase gene. J Biol Chem 270: 2640–2643, 1995
Skeido R, Murai K, Funahashi J, Fujisawa-Sehara A, Nabeshima Y, Kondoh H: The delta-crystallin enhancer-binding protein delta ef1 is a repressor of E2-box-mediated gene activation. Mol Cell Biol 14: 5692–5700, 1994
Chen CY, Schwartz RJ: Identification of novel DNA binding targets and regulatory domains of a murine tinman homeodomain factor, nkx-2.5. J Biol Chem 270: 15628–15633, 1995
Li P, He X, Gerrero MR, Mok M, Aggarwal A, Rosenfeld MG: Spacing and orientation of bipartite DNA-binding motifs as potential functional determinants for POU domain factors. Genes Dev 7: 2483–2496, 1993
Giguere V, Tini M, Flock G, Ong E, Evans RM, Otulakowski G: isoform-specific amino-terminal domains dictate DNA-binding properties of the ROR alpha, a novel family of orphan hormone nuclear receptors. Genes Dev 8: 538–553, 1994
Reisz-Porszasz S, Probst MR, Fukunaga BN, Hankinson O: Identification of functional domains of the aryl hydrocarbon receptor nuclear translocator protein (ARNT) Mol Cell Biol 14: 6075–6086, 1994
Harvath CM, Wen Z, Darnell JE Jr: A STAT protein domain that determines DNA sequence recognition suggests a novel DNA-binding domain. Genes Dev 9: 984–994, 1995
Bendall AJ, Molloym PI: Base preferences for DBA binding by the bHLH protein USF: Effects of mgcl2 on specificity and comparison with binding of myc family members. Nucl Acid Res 22: 2801–2810, 1994
Weintraub H, Davis R, Tapscott S, Thayer M, Krause M, Benezra R, Blackwell TK, Turner D, Rupp R, Hollenburg S, Zhuang Y, Lassar A: The myoD gene family: Nodal point during specification of the muscle cell lineage. Science 251: 761–766, 1991
Ayoubi TAY, Van de Ven WJM: Regulation of gene expression by alternative promoters. FASEB J 10: 453–460, 1996
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mhaskar, Y., Armour, G. & Dunaway, G. Alteration of the levels of the M-type 6-phosphofructo-1-kinase mRNA isoforms during neonatal maturation of heart, brain, and muscle. Mol Cell Biochem 214, 81–87 (2000). https://doi.org/10.1023/A:1007195017569
Issue Date:
DOI: https://doi.org/10.1023/A:1007195017569