Abstract
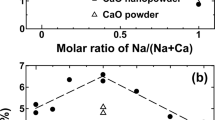
20%SrO-20%La2O3/CaO catalyst (SLC-2), prepared by impregnation, has shown 18% CH4 conversion and 80% C2-selectivity for the oxidative coupling of methane (OCM) at 1073–1103 K with CH4∶O2 molar ratio=9∶1 and total flow rate of 100 ml/min. Addition of SrO onto La2O3/CaO (LC) catalyst strengthens the surface basicity and leads to an increase in CH4 conversion and C2-selectivity. Meanwhile, the reaction temperature required to obtain the highest C2-yield increases with increasing SrO content. The formation of carbonate on the catalyst surface is the main reason for the deactivation of LC and SLC catalysts. If the amount of CO2 added into the feed is appropriate and the reaction temperature is high enough, there is no deactivation at all. In such case, the added CO2 will suppress the formation of CO2 produced via the OCM reaction, therefore, improves the C2-selectivity. The FT-IR spectra of CO2 adspecies recorded at different temperatures show that CO2 interacts easily with the catalyst surface to form different carbonate adspecies. Unidentate carbonate is the main CO2 adspecies formed on the catalyst surface. On the LC catalyst surface, the unidentate carbonate was first formed on Ca2+ cations at room temperature. If the temperature is higher than 473 K, it will form on La3+ cations. On the SLC catalyst surface, if the temperature is lower than 573 K, only the unidentate carbonate formed on Ca2+ cations could be observed. When the temperature is higher than 673 K, it will then form on Sr2+ cations. This suggests that the unidentate carbonate can migrate on the LC and SLC catalyst surface on one hand, and on the other hand, that the surface composition of SLC catalysts is dynamic in nature. On the basis of both the decomposition temperatures of the carbonate species, and the temperature dependence of the δΝ value which is the difference of symmetric and asymmetric stretching frequencies of surface carbonates, the in situ FT-IR technique offered two approaches to measure the surface basicity of the SLC catalyst. The results thus obtained are in good agreement with that of CO2-TPD. The role of the surface basicity of the SLC catalyst is also discussed.
Similar content being viewed by others
References
J.H. Lunsford, in:Natural Gas Conversion II, Studies in Surface Science and Catalysis, Vol. 81, eds. H.E. Curry-Hyde and R.F. Howe (Elsevier, Amsterdam, 1994)p. 1.
T. Ito, J.-X Wang, C.H. Lin and J.H. Lunsford, J. Am. Chem. Soc. 107 (1985) 5062.
D.J.C. Yates and N.F. Zlotin, J. Catal. 111 (1988) 317.
C.J. Cameron, Catal. Today 6 (1990) 324.
K. Aika and T. Nishiyama, J. Chem. Soc. Chem. Commun. (1988) 70.
J.A.S.P. Carreiro and M.J. Baerns, J. Catal. 117 (1989) 258.
K. Otsuaka, A.A. Said, K. Jinno and T. Komatsu, Chem. Lett. (1987) 77.
K.D. Campbell, H. Zhang, and J.H. Lunsford, J. Phys. Chem. 92 (1988) 750.
Y. Tong, M.P. Rosynek and J.H. Lunsford, J. Catal. 126 (1990) 291.
H. Yamashita, A. Kato, N. Watanabe and S. Matsuda, Nippon Kagaku Kaishi (1986) 1169.
S.J. Korf, J.A. Roos, N.A. de Bruijn, J.G.V. Ommen and J.R.H. Roos, J. Chem. Soc. Chem. Commun. (1987) 1433.
T. Suzuki, K. Wada and Y. Watanabe, Appl. Catal. 59 (1990) 213.
K. Aika and T. Nishiyama, Catal. Today 4 (1989) 271.
T. Nishiyama and K. Aika, J. Catal. 122 (1990) 346.
Y. Xu, L. Yu, J. Huang, W. Li and Z. Lin,Proc. Int. Conf. on Petrol. Refin. and Petrochem. Process., Beijing, 11–15 September 1991, p. 1373.
J.A. Lercher, C. Colombier and H. Noller, J. Chem. Soc. Faraday Trans. I 80 (1984) 949.
R. Philipp, K. Omata, A. Aoki and K. Fujimoto, J. Catal. 134 (1992) 422.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Xu, Y., Yu, L., Cai, C. et al. A study of the oxidative coupling of methane over SrO-La2O3/CaO catalysts by using CO2 as a probe. Catal Lett 35, 215–231 (1995). https://doi.org/10.1007/BF00807178
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00807178