Abstract
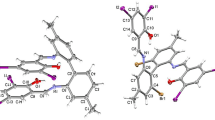
Three Schiff base ligands and their corresponding copper(II) complexes have been prepared and characterized. Elemental analyses, mass spectra, i.r., electronic spectra, μeff and the X- ray crystal structure for one of the complexes, as well as elemental analyses, mass spectra, i.r., electronic and 1H n.m.r. spectra of the ligands, have been obtained. The X-ray study shows that the geometry around the metal atom is intermediate between square planar and tetrahedral. Electrochemical studies on the complexes reveal a dependence of the CuII/CuI potentials on the extent of distortion of the planar structure observed in solid state.
Similar content being viewed by others
References
D. Chen, A. E. Martell and Y. Sun, Inorg. Chem., 28, 2647 (1989).
G. Henrici-Olive and S. OliveÂ, Angew. Chem. Int. Ed. Engl., 13, 29 (1971).
D. P. Kessissoglou, W. M. Butler and V. L. Pecoraro, Inorg. Chem., 26, 495 (1987).
D. E. Hamilton, R. S. Drago, and A. Zombeck, J. Am. Chem. Soc., 109, 374 (1987).
R. H. Holm and M. J. O' Connor, Prog. Inorg. Chem., 14, 241 (1971).
N. Matsumoto, Y. Nonaka, S. Kida, S. Kawano and I. Ueda, Inorg. Chim. Acta, 37, 27 (1979).
E. Acevedo-Arauz, J. M. Ferna ndez-G., M. J. Rosalez-Hoz and R. A. Toscano, Acta Crystallogr. Sect. C, C 48, 115 (1992).
M. L. Khorana and S. Y. Pandit, J. Indian Chem. Soc., 40, 789 (1963).
S. Patai, The Chemistry of the Carbon-Nitrogen Double Bond. Interscience Publishers. J Wiley, London, 1970. pp. 64±65.
S. Yamada, K. Kuge and K. Yamagouchi, Bull. Chem. Soc. Jpn., 40, 1864 (1967).
J. Heinze, Angew. Chem. Int. Ed. Engl., 23, 831 (1984).
W. R. Heineman and P. T. Kissinger. Laboratory Techniques in Electroanalytical Chemistry. Marcel Dekker. New York. 1984. Chap. 3.
D. T. Cromer and J. T. Waber. International Tables for X-Ray Crystallography, Vol. IV. Kynoch Press: Birmingham, England. 1974.
G. M. Sheldrick. SHELXTL/PC User's Manual. Siemens Analyt-ical X-rays Instruments, Inc. Madison Wisconsin. U.S.A. 1990.
M. Takayama, Y. Tanaka and T. Nomura, Org. Mass Spectrom., 28, 1529 (1993).
J. M. Ferna ndez-G., G. Espinosa-Pe rez, B. Quiro z-Garcõ a, R. A. Toscano and R. Salcedo. in preparation.
L. Sacconi and M. Ciampolini, J. Chem. Soc., 276 (1964).
J. E. Kovacic, Spectrocim. Acta, 23A, 183 (1967).
T. P. Cheesman, D. Hall and T. N. Waters, J. Chem. Soc. (A)_, 694 (1966).
A. L. Seligson and W. C. Trogler, J. Am. Chem. Soc., 113, 2520 (1991).
E. R. Brown and J. R. Sandifer in B. W. Rossiter and J. F. Hamilton (Eds.) Physical Methods of Chemistry. Electrochemical Methods. J Wiley, New York. 1986. Vol 2. Chap. 4.
W. E. Geiger, Prog. Inorg. Chem., 33, 275 (1985).
J. M. Ferna ndez-G., M. R. PatinÄ o-Maya, R. A. Toscano, L. Velasco, M. Otero-Lo pez and M. Aguilar-Martõ nez, Polyhedron, 16, 4371 (1997).
P. Zanello in I. Bernal (Ed.) Stereochemistry of Organometallic and Inorganic Compounds. Elsevier. Amsterdam. 1991. p. 180
R. S. Nicholson, Anal. Chem., 37, 1351 (1966).
J. M. Ferna ndez-G., M. J. Rosales-Hoz, M. F. Rubio-Arroyo, R. Salcedo, R. A. Toscano and A. Vela, Inorg. Chem., 26, 349 (1987).
G. S. Patterson and R. H. Holm, Bioinorg. Chem., 4, 257 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fernández-g, J.M., Acevedo-Arauz, E., Cetina-Rosado, R. et al. Electrochemical studies of copper(II) complexes derived from bulky Schiff bases. The crystal structure of bis[N-(1- adamantyl)-salicylaldiminato]copper(II). Transition Metal Chemistry 24, 18–24 (1999). https://doi.org/10.1023/A:1006923926389
Issue Date:
DOI: https://doi.org/10.1023/A:1006923926389