Summary
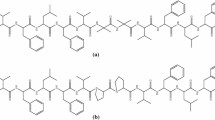
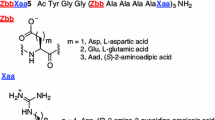
X-ray diffraction analyses have provided detailed structural information on the 310-helices of (i) pBrBz-d-(αMe)Phe-(Aib)2-d-(αMe)Phe-Aib-OtBu and Ac-(Aib)2-l-Lys(Bz)-(Aib)2-l-Lys(Bz)-(Aib)2-NHMe as suitable templates for molecular recognition studies, and (ii) pBrBz-TOAC-(l-Ala)2-TOAC-l-Ala-NHtBu as an appropriate spacer for an ESR study of side chain to side chain interactions. In addition, in Ac-TOAC-(Aib)2-l-Trp-Aib-OMe, forming a 310-helix, the TOAC residue plays the role of an effective quencher of the fluorescence of the tryptophan residue located one turn apart.
Similar content being viewed by others
References
Toniolo, C. and Benedetti, E., Macromolecules, 24 (1991) 4004.
Toniolo, C., Crisma, M., Formaggio, F., Valle, G., Cavicchioni, G., Précigoux, G., Aubry, A. and Kamphuis, J., Biopolymers, 33 (1993) 1061.
Toniolo, C., Formaggio, F., Crisma, M., Valle, G., Boesten, W.H.J., Schoemaker, H.E., Kamphuis, J., Temussi, P.A., Becker, E.L. and Précigoux, G. Tetrahedron, 49 (1993) 3641.
Toniolo, C., Bianco, A., Formaggio, F., Crisma, M. Bonora, G.M., Benedetti, E., DelDuca, V., Saviano, M., DiBlasio, B., Pedone, C. and Aubry, A., Bioorg. Med. Chem., 3 (1995) 1211.
Toniolo, C., Valente, E., Formaggio, F., Crisma, M., Pilloni, G., Corvaja, C., Toffoletti, A., Martinez, G.V., Hanson, M.P., Millhauser, G.L., George, C. and Flippen-Anderson, J.L., J. Pept. Sci., 1 (1995) 45.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Crisma, M., Bianco, A., Formaggio, F. et al. The polypeptide 310-helix as a template and a spacer. Lett Pept Sci 2, 187–189 (1995). https://doi.org/10.1007/BF00119149
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00119149