Abstract
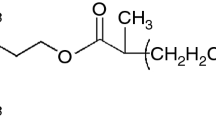
Fluorine and its derivatives have been used as a substitute to oxygen-containing oxidizers in higher performing propellants and other energetic materials. They typically react rapidly with metals or thin oxide layers of the metals to produce metal fluorides, generally in the gaseous state.
Thermal reactions of stoichiometric mixtures of metal (M=Al, Ti) -F containing polymers have been studied using thermal analyzers and their energetics are compared with the metals in other halogen element containing oxidizers. The experimental results indicated that thermal reaction becomes more exothermic as the bond strength between Al-X (X=F, Br, I) becomes stronger. It was also found that oxidation of metal powders is largely affected by the size of metal powders as well as the nature of existing oxide layer.
Similar content being viewed by others
References
S. F. Sarner, Propellant Chemistry, Chapter 9, Chapman & Hall, Ltd., London (1966) p. 217.
R. Reed, B. A. Zentner, C. D. Marrs and B. E. Mason, Energetics of the Fluorine Metal Reaction in Energetic Materials, Proc. 15th International Pyrotechnics, 1990 p. 815.
E. A. Turi, Thermal Characterization of Polymeric Materials, Academic Press, Inc., 1981 p. 221.
A. A. Shidlovskiy, Principles of Pyrotechnics, Mashinostroyeniye Press, 1973 p. 44.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, I., Reed, R.R., Brady, V.L. et al. Energy release in the reaction of metal powders with fluorine containing polymers. Journal of Thermal Analysis 49, 1699–1705 (1997). https://doi.org/10.1007/BF01983730
Issue Date:
DOI: https://doi.org/10.1007/BF01983730