Abstract
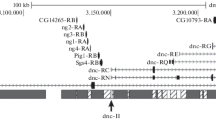
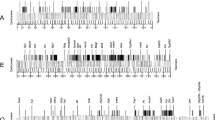
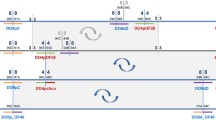
We have isolated a Hoppel-like transposon from heterochromatin of the second chromosome of Drosophila melanogaster and used a conserved DNA sequence between the different elements of this family to determine their distribution in both mitotic and polytene chromosomes. The hybridization pattern of polytene chromosomes extends throughout the entire chromocentre, as well as a substantial portion of the fourth chromosome. Analysis of different wild-type strains of D. melanogaster shows variation in euchromatic insertion sites, although most insertions are found near the chromocentre. The positions and the number of heterochromatic clusters of Hoppel on mitotic chromosomes are conserved among the several strains analysed. Accurate mapping of this element was achieved by in situ hybridization on D. melanogaster mitotic chromosomes that had previously been banded with Hoechst 33258. To evaluate the evolutionary stability of this pattern, different species were analysed by in situ hybridization and Southern blotting. We conclude that Hoppel has a conserved distribution in mitotic heterochromatin within the D. melanogaster subgroup, established around 5 million years ago. The overall conservation of heterochormatic organization supports the notion that heterochormatin does perform important structural and functional roles.
Similar content being viewed by others
References
Ashburner M (1989) Drosophila: A Laboratory Handbook and Manual. Cold Spring Harbor, New York: Cold Spring Harbor Laboratory Press.
Caizzi R, Caggese C, Pimpinelli S (1993) Bari-1, a new transposon-like family in Drosophila melanogaster with unique heterochromatic organization. Genetics 133: 335–345.
Carmena M and Gonzalez C (1995) Transposable elements map in a conserved pattern of distribution extending from β-heterochromatin to centromeres in Drosophila. Chromosoma 103: 676–684.
Carmena M, Abad J, Vilasante A, Gonzalez C (1993) The dodecassatellite sequence is closely linked to the centromere and can form connections between sister chromatids during mitosis. J Cell Sci 105: 41–50.
Coelho PA, Nurmisky D, Hartl D, Sunkel CE (1996) Identification of Porto-1: a new repeated sequence that localises close to the centromere of chromosome 2 of Drosophila melanogaster. Chromosoma 105: 211–222.
Dorer DR, Henikoff S (1994) Expansions of transgene repeats cause heterochromatin formation and gene silencing in Drosophila. Cell 77: 993–1002.
Frutos R, Peterson KR, Kidwell MG (1992) Distribution of Drosophila melanogaster transposable element sequences in species of the obscura group. Chromosoma 101: 293–300.
Gatti M, Pimpinelli S (1983) Cytological and genetical analysis of the Y chromosome of Drosophila melanogaster. Chromosoma 88: 349–373.
Gatti M, Pimpinelli S (1992) Functional elements in Drosophila melanogaster heterochromatin. Annu Rev Genet 26: 239–275.
Gonzalez C, Casal J, Ripoll P, Sunkel CE (1991) The spindle is required for the process of sister chromatid separation in Drosophila neuroblasts. Exp Cell Res 192: 10–15.
Hawley RS, Theurkauf WE (1993) Requiem for distributive segregation: aquiasmate segregation in Drosophila females. Trends Genet 9: 310–317.
Kellum R, Alberts BM (1995) Heterochromatin protein 1 is required for correct chromosome segregation in Drosophila embryos. J Cell Science 108: 1419–1431.
Khorodiliv NG, Bol'shakov VN, Blinov V, Solovév VV, Zhimulev IF (1987) Molecular genetic characterization of a new element in the Drosophila melanogaster genome with various localizations. Dokl Akad Nauk SSSR 295: 984–989.
Kurenova EV, Leibovitch BA, Bass IA, Bebikhov DV, Pavlova MN, Danilevskaya ON (1990) Hoppel-The family of Drosophila melanogaster mobile elements flanked by short inverted repeats and localised preferentially within the heterochromatic genomic regions. Genetica 26: 1701–1712.
Le M, Duricka D, Karpen GH (1995) Islands of complex DNA are widespread in Drosophila centric heterochromatin. Genetics 141: 283–303.
Lica L, Narayanswami S, Hamkalo B (1986) Mouse satellite DNA, centromere structure, and sister chromatid pairing. J Cell Biol 103: 1145–1151.
Locke J, McDermit HE (1993) Analysis of Drosophila chromosome 4 using pulse field gel electrophoresis. Chromosoma 102: 718–723.
Miklos GL, Yamamoto MT, Davies J, Pirrotta V (1988) Micro-cloning reveals a high frequency of repetitive sequences characteristic of chromosome 4 and the b-heterochromatin of Drosophila melanogaster. Proc Natl Acad Sci USA 85: 2051–2055.
Pimpinelli S, Dimitri P (1989) Cytogenetic analysis of segregation distortion in Drosophila melanogaster: the cytological organization of the Responder (Rsp) locus. Genetics 121: 765–772.
Pimpinelli S, Bonaccorsi S, Gatti M, Sandler L (1986) The peculiar genetic organization of Drosophila heterochromatin. Trends Genet 2: 17–20.
Pimpinelli S, Berloco M, Fanti L et al. (1995) Transposable elements are stable structural components of Drosophila melanogaster heterochromatin. Proc Natl Acad Sci USA 92: 3804–3808.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Coelho, P.A., Queiroz-machado, J., Hartl, D. et al. Pattern of Chromosomal Localization of the Hoppel Transposable Element Family in the Drosophila melanogaster Subgroup. Chromosome Res 6, 385–396 (1998). https://doi.org/10.1023/A:1009277322626
Issue Date:
DOI: https://doi.org/10.1023/A:1009277322626