Abstract
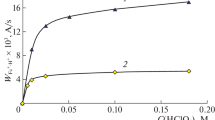
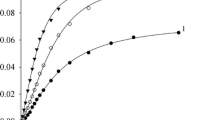
Ionization of 2- and 3-nitroanilines was studied in HCl-DMF-1,1,2,2-tetrachloro-ethane (TCE) solutions at 25 °C. The ionization capability of the medium and basicity constants pK i of indicators change depending on the ratio of the components. The numerical values of pK i are found to depend on the analytical composition of the DMF-TCE solvent. The solvent effect on pK i is associated with a change in the solvation of the nonionized form of the indicators.
Similar content being viewed by others
References
M. I. Vinnik,Usp. Khim., 1966,35, 1922 [Russ. Chem. Rev., 1966,35 (Engl. Transi.)].
M. I. Vinnik, I. S. Kislina, and N. B. Librovich,Dokl. Akad. Nauk SSSR, 1980,251, 138 [Dokl. Chem., 1980,251 (Engl. Transi.)].
C. H. Rochester,Acidity Functions, Acad. Press, London New York, 1970, Ch. 6.
S. G. Sysocva, I. S. Kislina, and M. I. Vinnik,Izv. Akad. Nauk SSSR, Ser. Khim., 1987,989 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1987,36, 909 (Engl. Transl.)].
I. S. Kislina, S. G. Sysocva, and O. N. Temkin. Izv. Akad. Nauk, Ser. Khim., 1994, 1025 [Rum Chem. Bull., 1994,43, 960 (Engl. Transl.)].
V. D. Maiorov, S. G. Sysocva, O. N. Temkin, and I. S. Kislina,Izv. Akad. Nauk, Ser. Khim., 1993, 1577 [Russ. Chem. Bull. 1993,42, 1511 (Engl. Transl.)].
I. S. Kislina, S. G. Sysocva, and O. N. Temkin,Izv. Akad. Nauk, Ser. Khim., 1990, 77 [Russ. Chem. Bull., 1996,45, 69 (Engl. Transl.)].
M. I. Vinnik,Kinet. Katal., 1987,28, 100 [Kinet. Catal., 1987,28 (Engl. Transl.)].
S. Janardhanan and C. Kalidas,Indian J. Chem., 1983,22A, 17.
Author information
Authors and Affiliations
Additional information
Translanted fromIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 6, pp. 1446–1449, June, 1996.
Rights and permissions
About this article
Cite this article
Kislina, I.S., Sysoeva, S.G. & Temkin, O.N. Solvent effect on catalytic properties of the HCI-DMF-1,1,2,2-tetrachloroethane system. Russ Chem Bull 45, 1376–1379 (1996). https://doi.org/10.1007/BF01434215
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01434215