Abstract
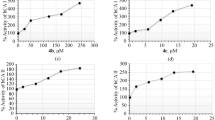
Biological activity of cholinesterases can be determined by optically monitoring the enzymatic reaction with indophenyl acetate, (N-4′-acetoxyphenyl)-4-quinone imine. At pH 8.0 cholinesterases hydrolyze this yellow dye to yield a blue reaction product. Cholinesterase inhibitors reduce the rate of this hydrolysis. Thus, by monitoring absorbance of the hydrolysis product at its maximum (630 nm) as a function of time, reaction rates of both cholinesterase activity and cholinesterase inhibition may be quantified spectroscopically. Using this technique, we measured the enzymatic activity of butyrylcholinesterase (BuChE) molecules encapsulated in tetramethyl orthosilicate (TMOS) silicate gel-glass prepared by hydrolysis and condensation. This activity is reduced, in a concentration-dependent manner, by the reversible cholinesterase inhibitors 1,5-bis(4-allyldimethyl-ammoniumphenyl) pentan- 3-one dibromide (BADAPP) and 9-amino-1,2,3,4-tetrahydroacridine (THA; tacrine, Cognex). The gel-glasses are rigid and compact, transparent, and porous enough to allow reagents to diffuse in and out.
Similar content being viewed by others
References
D. Avnir, S. Braun, and M. Ottolenghi in Supramolecular Architecture: Synthetic Control in Thin Films and Solids, edited by T. Bein (ACS Symposium Series #499, 1992).
J.C. Pouxviel, B. Dunn, and J.I. Zink, J. Phys. Chem. 93, 2134 (1989).
R. Reisfeld, J. Non-Cryst. Solids 121, 254 (1990).
A. Slama-Schwok, D. Avnir, and M. Ottolenghi, J. Phys. Chem. 93, 7544 (1989).
C.J. Brinker and G. Scherer, Sol-Gel Science: The Physics and Chemistry of Sol-Gel Processing (Academic Press, San Diego, 1990), p. 601.
S.A. Yamanaka, B.S. Dunn, J.S. Valentine, and J.I. Zink, J. Am. Chem. Soc. 117, 9095 (1995).
S.A. Yamanaka, N.P. Nguyen, L.M. Ellerby, B.S. Dunn, J.S. Valentine, and J.I. Zink, J. Sol-Gel Sci. and Tech. 2, 827 (1994).
L.M. Ellerby, C.R. Nishida, F. Nishida, S.A. Yamanaka, B. Dunn, J.S. Valentine, and J.I. Zink, Sci. 255, 1113 (1992).
S. Braun, S. Rappoport, R. Zusman, D. Avnir, and M. Ottolenghi, Mat. Lett. 10, 1 (1990).
S. Braun, S. Rappoport, S. Shtelzer, R. Zusman, S. Druckman, D. Avnir, and M. Ottolenghi, in Biotech: Bridging Research and Applications, edited by D. Kanely (Kluwer Academic Publishers, Boston, 1991).
D. Avnir, S. Braun, O. Lev, and M. Ottolenghi, Chem. Mat. 6, 1605 (1994).
B. Dave, B. Dunn, J.S. Valentine, and J.I. Zink, Anal. Chem. 66, 1120A (1994).
E.K. Perry, Br. Med. Bull. 42, 63 (1986).
C.Q. Mountjoy, M.N. Rossor, and N.R. Evans et al., Biol. Psychiatry, 1415 (1986).
W.K. Summers, L.V. Majovski, G.M. Marsh, K. Tachiki, and A. Kling, New Engl. J. Med. 26, 257 (1986).
L.J. Fitten, K.M. Perryman, P.L. Gross, H. Fine, J. Cummins, and C. Marshall, Am. J. Psychiatr. 147, 239 (1990).
D.N. Kramer and R.M. Gamson, Anal. Chem. 30, 251 (1958).
G.K. Huber, in Methods of Enzymatic Analysis, 3rd ed., Vol. XII: Drugs and Pesticides, edited by Bergmeyer (VCH, 1986), p. 412.
Rights and permissions
About this article
Cite this article
Akbarian, F., Lin, A., Dunn, B. et al. Spectroscopic Determination of Cholinesterase Activity and Inhibition in Sol-Gel Media. Journal of Sol-Gel Science and Technology 8, 1067–1070 (1997). https://doi.org/10.1023/A:1018350828807
Issue Date:
DOI: https://doi.org/10.1023/A:1018350828807