Abstract
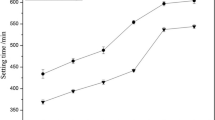
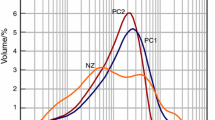
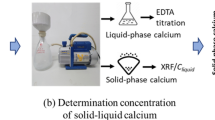
The pH of a hydraulic calcium phosphate cement (HCPC) made of monocalcium phosphate monohydrate (Ca(H2PO4)2·H2O; MCPM), β-tricalcium phosphate (β-(Ca3(PO4)2; β-TCP) and water was measured as a function of reaction time and composition at room temperature. During setting, the cement pH varies from very acidic pH values, i.e., 2.5, to almost neutral pH values, i.e., 6. The cement pH profile significantly depends on the initial cement composition. However, all profiles are characterized by a sharp initial decrease of the pH due to the dissolution of MCPM crystals and the precipitation of dicalcium phosphate dihydrate (CaHPO4· 2H2O; DCPD) crystals. With an excess of MCPM, the final pH stays low, and its value can be predicted from the initial composition of the cement and solubility data. With an excess of β-TCP, the end pH is close to 5, which is much lower than 5.9, the value predicted by calculation. Results suggest that the difference may be due to the presence of impurities in the cement. Replacing MCPM by phosphoric acid renders the cement paste very acidic for the initial 30 s, but then the pH profile follows that obtained with MCPM. Adding pyrophosphate ions into the cement paste postpones the position of the pH minimum. The delay, which is proportional to the concentration of pyrophosphate ions, is thought to be due to the inhibiting action of pyrophosphate ions on the precipitation of DCPD crystals.
Similar content being viewed by others
References
P. D. COSTANTINO, C. D. FRIEDMAN, K. JONES, L. C. CHOW, H. J. PELZER and G. A. SISSON, Arch. Otolaryngol. Head Neck Surg. 117 (1991) 379.
C. D. FRIEDMAN, P. D. COSTANTINO, K. JONES, L. C. CHOW, H. J. PELZER and G. A. SISSON, ibid. 117 (1991) 385.
M. L. SHINDO, P. D. COSTANTINO, C. D. FRIEDMAN and L. C. SHOW, ibid 119 (1993) 185.
D. B. KAMERER, B. E. HIRSCH, C. H. SNYDERMAN, P. D. COSTANTINO and C. D. FRIEDMAN, Amer. J. Otology 15 (1994) 47.
B. R. COSTANTZ, I. C. ISON, M. T. FULMER, R. D. POSER, S. T. SMITH, M. VANWAGONER, J. ROSS, S. A. GOLDSTEIN, J. B. JUPITER and D. I. ROSENTHAL, Science 267 (1995) 1796.
E. MUNTING, A. A. MIRTCHI and J. LEMAITRE, J. Mater. Sci. Mater. Med. 4 (1993) 337.
K. OHURA, M. BOHNER, P. HARDOUIN, J. LEMAITRE, G. PASQUIER and B. FLAUTRE, J. Biomed. Mater. Res. 30 (1996) 193.
A. A. MIRTCHI, J. LEMAITRE and N. TERAO, Biomaterials 10 (1989) 475.
M. BOHNER, J. LEMAITRE and T. A. RING, in Proceedings of the 3rd Euro-Ceramics Conference, Madrid, September 1993, edited by P. Durán and J. F. Fernández (Faenza Editrice Iberica, Agenda del Rey, Spain, 1993) p. 95.
M. BOHNER, PhD thesis No. 1171, Swiss Federal Institute of Technology (EPFL), Lausanne, (1993).
M. BOHNER, J. LEMAITRE and T. A. RING, J. Amer. Ceram. Soc. 79 (1996) 1427.
F. L. L. B. CARNEIRO and A. BARCELLOS, Union Testing Res. Lab. Mater. Structures 13 (1953) 97.
G. VEREECKE and J. LEMAITRE, J. Crystal Growth 104 (1990) 820.
R. W. MARSHALL and G. H. NANCOLLAS, J. Phys. Chem. 73 (1969) 3838.
T. V. VASUDEVAN, P. SOMASUNDARAN, C. L. HOWIE-MEYERS, D. L. ELLIOT and K. P. ANANTHAPADMANABHAN, Langmuir 10 (1994) 320.
M. BOHNER, J. LEMAITRE and T. A. RING, J. Colloid Interface Sci. (in press).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BOHNER , M., VAN LANDUYT , P., MERKLE , H.P. et al. Composition effects on the pH of a hydraulic calcium phosphate cement. Journal of Materials Science: Materials in Medicine 8, 675–681 (1997). https://doi.org/10.1023/A:1018583706335
Issue Date:
DOI: https://doi.org/10.1023/A:1018583706335