Abstract
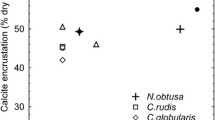
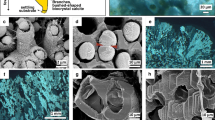
Calcite precipitation in spring 1988 in Lake Constance was studied by measuring physical, chemical and biological parameters in daily intervals. Algae triggering calcite precipitation in spring after an induction period of 1 day wereStephanodiscus hantzschii andChlorella spp. whereas no time lag concerning maximum saturation index was observed. Number and size distribution of crystals were determined by microscopical methods. Simple equations were given for estimation of the precipitated mass of calcite by approximating the volume of calcite crystals by simple geometric bodies and multiplying by crystal numbers. Particulate calcium was analysed after dissolving suspended matter and amounted to 90% of inorganic dry weight. Crystals were observed for the first time after dissolved phosphorus was no more detectable.
Similar content being viewed by others
References
Bischoff, J. L., 1968. Kinetics of calcite nucleation: magnesium ion inhibition and ionic strength catalysis. J. Geophys. Res. 73:3315–3322.
Bodine, M. W., H. D. Holland and M. Borcsik, 1965. Coprecipitation of manganese and strontium with calcite. Symposium: Problems of postmagm. ore depos. 2:401–406.
Braunwarth, C., 1988. Populationsdynamik natürlicher Phyto-planktonpopulationen: Analyse der In-Situ Wachstums- und Verlustraten. Dissertation Univ. Konstanz, Vol. 209. Konstanz: Hartung Gorre Verlag.
Brunskill, G. J., 1969. Fayatteville Green Lake, New York II. Precipitation and sedimentation of calcite in a meromictic lake with laminated sediments. Limnol. Oceanogr. 14:858–861.
Carter, P. W., 1978. Adsorption of amino acid-containing organic matter by calcite and quartz. Geochim. Cosmochim. Acta 42:1239–1242.
Carter, P. W., R. M. Mitterer, 1978. Amino acid composition of organic matter associated with carbonate and noncarbonate sediments. Geochim Cosmochim. Acta 42:1231–1238.
Chave, K. E., 1965. Carbonates: association with organic matter in surface seawater. Science 148: 1723–1724.
Chave, K. E., E. Suess, 1970. Calcium carbonate saturation in seawater: effects of dissolved organic matter. Limnol. Oceanogr. 15:633–637.
Curtis, G. P., M. Reinhard, P. V. Roberts, 1986. Sorption of hydrophobic organic compounds by sediments. In: J. A. Davis, K. F. Hayes (eds), Geochemical processes at mineral surfaces. Americal Chemical Society, pp. 191–216.
De Groot, K., E. M. Duyvis, 1966. Crystal form of precipitated calcium carbonate as influenced by adsorbed magnesium ions. Nature 8:183–184.
Effler, S. W., 1984. Carbonate equilibria and the distribution of inorganic carbon in Saginaw Bay. J. Great Lakes Res. 10:3–14.
Effler, S. W., C. T. Driscoll, 1985. Calcium chemistry and deposition in ionically enriched Onondaga Lake, New York. Environ. Sci. Technol. 19:716–720.
Effler, S. W., S. D. Field, D. A. Wilcox, 1981. The carbonate chemistry of Green Lake, Jamesville, New York. J. Freshwat. Ecol. 1:141–153.
Effler, S. W., S. D. Field, M. Quirk, 1982. The seasonal cycle of inorganic carbon species in Cazenovia Lake, New York, 1977. Freshwat. Biol 12:139–147.
Folk, R. L., 1974. The natural history of crystalline calcium carbonate: effect of magnesium content and salinity. J. Sed. Petrol. 44:40–53.
Freeman, J. S., D. L. Rowell, 1981. The adsorption and precipitation of phosphate onto calcite. J. Soil Sci. 32:75–84.
Garofalo, J. E., S. W. Effler, 1987. Coating of zooplankton with calcium in Onondaga Lake, New York. Environ. Sci. Technol. 21:604–606.
Giannimaras, E. K., P. G. Koutsoukos, 1987. The crystallization of calcite in the presence of orthophosphate. J. Colloid Interf. Sci. 116:423–430.
Green, W. J., D. E. Canfield, B. A. Steinly, 1985. Special variations in and controls on the calcite saturation index in Acton Lake, Ohio. Freshwat. Biol. 15:525–533.
Green, D. B., T. J. Logan, N. E. Smeck, 1978. Phosphate adsorption-desorption characteristics of suspended sediments in the Maumee River basin of Ohio. J. Environ. Qual. 7:208–212.
Holland, H. D., M. Borscik, J. Munoz, U. M. Oxburgh, 1963. The coprecipitation of Sr2+ with aragonite and of Ca2+ with strontianite between 90 °C and 100 °C. Geochim. Cosmochim. Acta 27:957–977.
House, W. A., 1987. Inhibition of calcite crystal growth by inorganic phosphate. J. Colloid Interf. Sci. 119:505–511.
House, W. A., L. Donaldson, 1986. Adsorption and coprecipitation of phosphate on calcite. J. Colloid Interf. Sci. 112:309–324.
House, W. A., H. Casey, L. Donaldson, S. Smith, 1986. Factors affecting the coprecipitation of inorganic phosphate with calcite in hardwaters. Wat. Res. 20:917–922.
Jurinak, J. J., R. A. Griffin, 1972. Nitrate ion adsorption by calcium carbonate. Soil Sci. 113:130–135.
Kelts, K., K. J. Hsu, 1978. Freshwater carbonate sedimentation. In: A. Lerman (ed.) Lakes, chemistry, geology and physics, New York: Springer, pp. 295–324.
Kilham, S. S., 1984. Silicon and phosphorus growth kinetics and competitive interactions betweenStephanodiscus minutus andSynedra sp. Verh. Internat. Verein. Limnol. 22:435–439.
Kinsman, D. J. J., H. D. Holland, 1969. The coprecipitation of cations with CaCO3. IV. The coprecipitation of Sr2+ with aragonite between 16 °C and 96 °C. Geochim. Cosmochim. Acta 33:1–17.
Kitano, Y., D. W. Hood, 1965. The influence of organic material on the polymorphic crystallization of calcium carbonate. Geochim. Cosmochim. Acta 29:29–41.
Kitano, Y., M. Okumura, M. Idogaki, 1978. Uptake of phosphate ions by calcium carbonate. Geochem. J. 12:29–37.
Kitano, Y., M. Okumura, M. Idogaki, 1979. Behavior of dissolved silica in parent solution at the formation of calcium carbonate. Geochem. J. 13:253–260.
Kleiner, J., 1988. Coprecipitation of phosphate with calcite in lake water: a laboratory experiment modelling phosphorus removal with calcite in Lake Constance. Wat. Res. 22:1259–1265.
Koschel, R., J. Benndorf, G. Proft, F. Recknagel, 1983. Calcite precipitation as a natural control mechanism of eutrophication. Arch. Hydrobiol. 98:380–408.
Koschel, R., G. Proft, H. Raidt, 1987. Autochthone Kalkfällung in Hartwasserseen der Mecklenburger Seenplatte. Limnologica 18:317–338.
Küchler-Krischun, J., (in preparation) Calcite precipitation induced by phytoplanktonic algae.
Kunz, B., 1983. Heterogene Nukleierung und Kristallwachstum von CaCO3 (Calcit) in natürlichen Gewässern. Ph. D. Thesis, ETH Zürich.
Lahann, R. W., R. C. Campbell, 1980. Adsorption of palmitic acid on calcite. Geochim. Cosmochim. Acta 44:629–634.
Lee, C., J. McKenzie, M. Sturm, 1987. Carbon isotope fractionation and changes in the flux and composition of particulate matter resulting from biological activity during a sediment trap experiment in Lake Greifen, Switzerland. Limnol. Oceanogr. 32:83–96.
Lorens, R. B., 1981. Sr, Cd, Mn and Co distribution coefficients in calcite as a function of calcite precipitation rate. Geochim. Cosmochim. Acta 45:553–561.
Lund, J. W. G., C. Kipling, E. D. Le Cren, 1958. The inverted microscope method for estimating algal numbers and the statistical basis of estimations by counting. Hydrobiologia 11:143–170.
Megard, R. O., 1968. Planktonic photosynthesis and the environment of calcium carbonate deposition in lakes. Interim Report Limnol. Res. Center Univ. Minnesota 2:1–47.
Minder, L., 1922. Über biogene Entkalkung im Zürichsee. Verh. Internat. Verein Limnol. 1:20–32.
Möller, P., G. Rajagopalan, 1975. Precipitation kinetics of CaCO3 in presence of Mg2+ ions. Zeitschr. Phys. Chem. Neue Folge 94:297–314.
Mucci, A., J. W. Morse, 1983. The incorporation of Mg2+ and Sr2+ into calcite overgrowth: influences of growth rate and solution composition. Geochim. Cosmochim. Acta 47:217–233.
Müller, G., 1969. Sedimentbildung im Plattensee/Ungarn. Naturwiss. 12:606–615.
Müller, G., 1970. High-magnesian calcite and protodolomite in Lake Balaton (Hungary) sediments. Nature 226:749–750.
Müller, G., 1971. Aragonite inorganic precipitation in a freshwater lake. Nature Phys. Sci. 229:18.
Murphy, T. P., K. J. Hall, I. Yesaki, 1983. Coprecipitation of phosphate with calcite in a naturally eutrophic lake. Limnol. Oceanogrw. 28:58–69.
Niessen, F., M. Sturm, 1987. Die Sedimente des Baldeggersees (Schweiz) — Ablagerungsraum und Eutrophierungsentwicklung während der letzten 100 Jahre. Arch. Hydrobiol. 108:365–383.
Ohle, W., 1952. Die hypolimnische Kohlendioxid-Akkumulation als produktionsbiologischer Indikator. Arch. Hydrobiol. 46:153–285.
Okumura, M., Y. Kitano, M. Idogaki, 1983. Incorporation of fluoride into calcite — effects of organic materials and magnesium ions in a parent solution. Geochem. J. 17:157–263.
Oomori, T., Y. Kitano, 1985. Catalytic effect of magnesium ions on polymorphic crystallization on calcium carbonate. Bull. College Sci. Univ. Ryukyus 39:57–62.
Osman-Sigg, G., W. Stumm, 1982. Partikelgrößenverteilung und natürliche Koagulation im Zürichsee. Schweiz. Z. Hydrol. 44:405–421.
Otsuki, A., R. G. Wetzel, 1972. Coprecipitation of phosphate with carbonates in a marl lake. Limnol. Oceanogr. 17:763–767.
Otsuki, A., R. G. Wetzel, 1973. Interaction of yellow organic acids with calcium carbonate in freshwater. Limnol. Oceanogr. 18:490–493.
Otsuki, A., R. G. Wetzel, 1974. Calcium and total alkalinity budgets and calcium carbonate precipitation of a small hardwater lake. Arch. Hydrobiol. 73:14–30.
Pingitore, N. E., 1986. Modes of coprecipitation of Ba2+ and Sr2+ with calcite. In: J. A. Davis, K. F. Hayes (eds.), Geochemical processes at mineral surfaces, chapter 27, Washington: American Chemical Society, pp. 574–586.
Pingitore, N. E., M. P. Eastman, 1986. The coprecipitation of Sr2+ with calcite at 25 °C and 1 atm. Geochim. Cosmochim. Acta 50:2195–2203.
Plummer, L. N., E. Busenberg, 1982. The solubilities of calcite, aragonite and vaterite in CO2-H2O solutions between 0 and 90 °C, and an evaluation of aqueous model for the system CaCO3-CO2-H2O. Geochim. Cosmochim. Acta 46:1011–1040.
Portner, C., 1951. Le mécanisme de la précipitation du CO3Ca et la détermination de l'épaisseur du dépot annuel de la vase dans le lac du Neuchatel. Schweiz. Z. Hydrol. 13:199–290.
Reddy, M. M., G. H. Nancollas, 1973. Calcite crystal growth inhibition by polyphosphonates. Desalination 12:61–73.
Reynolds, R. C., 1978. Polyphenol inhibition of calcite precipitation in Lake Powell. Limnol. Oceanogr. 23:585–597.
Roßknecht, H., 1977. Zur autochthonen Calcitfällung im Bodensee-Obersee. Arch. Hydrobiol. 81:35–64.
Roßknecht, H., 1980. Phosphateliminierung durch autochthone Calcitfällung im Bodensee-Obersee. Arch. Hydrobiol. 88:328–344.
Rudek, R., H. Sontheimer, 1976. Einfluß natürlicher Wasserinhaltsstoffe auf die Ausfallge-schwindigkeit von CaCO3. Vom Wasser 47:421–431.
Sapozhnikov, D. G., A. A. Tsvetkov, 1959. Separation of hydrated calcium carbonate from the bottom of Lake Issyk-Kul. Dokl. AN SSSR 124:2. Cited in: E. I. Semenov, 1964, Hydrated carbonates of sodium and calcium. Soviet Phys. Cryst. 9:88–90.
Schroeder, J. H., 1969. Experimental dissolution of calcium, magnesium, and strontium from recent biogenic carbonates: a model of diagenesis. J. Sed. Petrol. 39:1057–1073.
Schroeder, H. G., J. Schneider, 1980. Hydrochemische Untersuchungen zum Karbonatkreislauf des Attersees (Salzkammergut, Österreich). Arb. Lab. Weyregg 4:235–257.
Schroeder, H. G., H. Windolph, J. Schneider, 1983. Bilanzierung der biogenen Karbonatproduktion eines oligotrophen Sees (Attersee, Salzkammergut — Österreich). Arch. Hydrobiol. 97:356–372.
Sigg, L., M. Sturm, D. Kistler, 1987. Vertical transport of heavy metals by settling particles in Lake Zürich. Limnol. Oceanogr. 32:112–130.
Sommer, U., 1985. Seasonal sucsession of phytoplankton in Lake Constance. Bio Science 35:351–357.
Stabel, H. H., 1985. Mechanisms controlling the sedimentation sequence of various elements in prealpine lakes. In: W. Stumm (ed.), Chemical processes in lakes, New York: Wiley Interscience Publ., pp. 143–167.
Stabel, H. H., 1986a. Calcite precipitation in Lake Constance: chemical equilibrium, sedimentation, and nucleation by algae. Limnol. Oceanogr. 31:1081–1093.
Stabel, H. H., 1986b. The role of plankton biomass in controlling fluctuations of suspended matter in Lake Constance. Hydrobiologia 140:173–181.
Stabel, H. H., 1987. Settling velocity and residence time of particles in Lake Constance. Swiss J. Hydrol. 49:284–293.
Stabel, H. H., 1988. Algal control of elemental sedimentary fluxes in Lake Constance. Verh. Internat. Verein. Limnol. 23:700–706
Stabel, H. H., 1989. Coupling of strontium and calcium cycles in Lake Constance. Hydrobiologia 176/177:323–329.
Stabel, H. H., C. Chondrogianni, 1988. Seasonal shifts in grain size of settling particles in Lake Constance. Wat. Res. 22:251–255.
Stabel, H. H., J. Kleiner, 1983. Endogenic flux of manganese to the bottom of Lake Constance. Arch. Hydrobiol. 98:307–316.
Stabel, H. H., J. Küchler-Krischun, J. Kleiner, P. Merkel, 1986. Removal of strontium from Lake Constance water by coprecipitation with calcite. Naturwiss. 73:551–553.
Stewart, A. J., R. G. Wetzel, 1981. Dissolved humic materials: photodegradation, sediment effects, and reactivity with phosphate and calcium carbonate precipitation. Arch. Hydrobiol. 92:265–286.
Stoffers, P., R. Fischbeck, 1974. Monohydrocalcite in the sediments of Lake Kivu (East Africa). Sedimentology 21:163–170.
Sturm, M., 1985. Schwebstoffe in Seen — Particules en suspension dans les lacs. Mitt./Nouv. EAWAG 19:9–15.
Sturm, M., U. Zeh, J. Müller, L. Sigg, H. H. Stabel, 1982. Schwebstoffuntersuchungen im Bodensee mit Intervall-Sedimentationsfallen. Eclogae geol. Helv. 75:579–588.
Strong, A. E., B. J. Eadie, 1978. Satellite observations of calcium carbonate precipitation in the Great Lakes. Limnol. Oceanogr. 23:877–887.
Suess, E., 1970. Interaction of organic compounds with calcium carbonate. I. Association phenomena and geochemical implications. Geochim. Cosmochim. Acta 34:157–168.
Suess, E., 1973. Interaction of organic compounds with calcium carbonate. II. Organocarbonate association in recent sediments. Geochim. Cosmochim. Acta 37:2435–2447.
Taylor, G. F., 1972. The occurrence of monohydrocalcite in two small lakes in the southeast of south australia. Am. Mineral. 60:690–697.
Tilzer, M. M., B. Beese, 1988. The seasonal productive cycle of phytoplankton and controlling factors in Lake Constance. Schweiz. J. Hydrol. 50:1–39.
Utermöhl, H., 1958. Zur Vervollkommnung der quantitativen Phytoplankton-Methodik. Int. Verein. Theor. Angew. Limnol. 9:1–39.
Vanderploeg, H. A., B. J. Eadie, J. R. Liebig, S. J. Tarapchak, 1987. Contribution of calcite to the particle-size spectrum of Lake Michigan seston and its interactions with the plankton. Can. J. Fish. Aquat. Sci. 44:1898–1914.
Weidemann, A. D., T. T. Bannister, S. W. Effler, D. L. Johnson, 1985. Paritculate and optical properties during CaCO3 precipitation in Otisco Lake. Limnol. Oceanogr. 30:1078–1083.
Weilenmann, U., C. O'Melia, W. Stumm, 1989. Particle transport in lakes: models and measurements. Limnol. Oceanogr. 34:1–18.
Wetzel, R. G., H. L. Allen, 1970. Functions and interactions of dissolved organic matter and the littoral zone in lake metabolism and eutrophication. Proceedings IBP-UNESCO Symp. Prod. Probl. Freshwat., pp. 334–347.
Wetzel, R. G., W. S. White, 1985. Alteration of iron-CaCO3 precipitation by yellow organic acids of aquatic angiosperm origin. Arch. Hydrobiol. 104:247–251.
White, W. S., R. G. Wetzel, 1985. Association of vitamin B12 with calcium carbonate in hardwater lakes. Arch. Hydrobiol. 104:305–309.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Küchler-Krischun, J., Kleiner, J. Heterogeneously nucleated calcite precipitation in Lake Constance. A short time resolution study. Aquatic Science 52, 176–197 (1990). https://doi.org/10.1007/BF00902379
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00902379