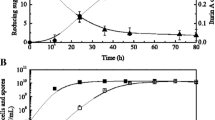
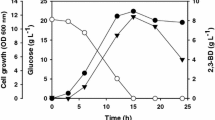
Abstract
Two recombinants of alkaliphilic Bacillus subtilis LOCK 1086, constructed via different strategies such as cloning the gene encoding bacterial hemoglobin from Vitreoscilla stercoraria (vhb) and overexpression of the gene encoding acetoin reductase/2,3-butanediol dehydrogenase (bdhA) from B. subtilis LOCK 1086, did not produce more 2,3-butanediol (2,3-BD) than the parental strain. In batch fermentations, this strain synthesized 9.46 g/L in 24 h and 12.80 g/L 2,3-BD in 46 h from sugar beet molasses and an apple pomace hydrolysate, respectively. 2,3-BD production by B. subtilis LOCK 1086 was significantly enhanced in fed-batch fermentations. The highest 2,3-BD concentration (75.73 g/L in 114 h, productivity of 0.66 g/L × h) was obtained in the sugar beet molasses–based medium with four feedings with glucose. In a medium based on the apple pomace hydrolysate with three feedings with sucrose, B. subtilis LOCK 1086 produced up to 51.53 g/L 2,3-BD (in 120 h, productivity of 0.43 g/L × h).






Similar content being viewed by others
References
Alam S, Capit F, Weigandg WA, Hong J (1990) Kinetics of 2,3-butanediol fermentation by Bacillus amyloliquefaciens: effect of initial substrate concentration and aeration. J Chem Tech Biotechnol 41:71–84. doi:10.1002/jctb.280470109
Barham D, Trinder P (1972) An improved color reagent for the determination of blood glucose by the oxidase system. Analyst 97:142–145. doi:10.1039/AN9729700142
Biswas R, Yamaoka M, Nakayama H, Kondo T, Yoshida K, Bisaria VS, Kondo A (2012) Enhanced production of 2,3-butanediol by engineered Bacillus subtilis. Appl Microbiol Biotechnol 94:651–658. doi:10.1007/s00253-011-3774-5
Budsławski J, Drabant Z (1972) Food analysis methods. WNT, Warsaw
Celińska E, Grajek W (2009) Biotechnological production of 2,3-butanediol—current state and prospects. Biotechnol Adv 27:715–725. doi:10.1016/j.biotechadv.2009.05.002
Cheng KK, Liu Q, Zhang JA, Li JP, Xu JM, Wang GH (2010) Improved 2,3-butanediol production from corncob acid hydrolysate by fed-batch fermentation using Klebsiella oxytoca. Process Biochem 45:613–616. doi:10.1016/j.procbio.2009.12.009
Dai JY, Zhao P, Cheng XL, Xiu ZL (2015) Enhanced production of 2,3-butanediol from sugarcane molasses. Appl Biochem Biotechnol 175:3014–3024. doi:10.1007/s12010-015-1481-x
Gao J, Xu H, Li Q, Feng X, Li S (2010) Optimization of medium for one-step fermentation of inulin extract from Jerusalem artichoke tubers using Paenibacillus polymyxa ZJ-9 to produce R,R-2,3-butanediol. Bioresour Technol 101:7076–7082. doi:10.1016/j.biortech.2010.03.143
Geckil H, Barak Z, Chipman DM, Erenler SO, Webster DA, Stark BC (2004) Enhanced production of acetoin and butanediol in recombinant Enterobacter aerogenes carrying Vitreoscilla hemoglobin gene. Bioprocess Biosyst Eng 26:325–330. doi:10.1007/s00449-004-0373-1
Guo X, Cao Ch, Wang Y, Li Ch, Wu M, Chen Y, Zhang C, Pei H, Xiao D (2014) Effect of the inactivation of lactate dehydrogenase, ethanol dehydrogenase, and phosphotransacetylase on 2,3-butanediol production in Klebsiella pneumoniae strain. Biotechnol Biofuels 7:44. doi:10.1186/1754-6834-7-44
Huang ChF, Jiang YF, Guo GL, Hwang WS (2013) Method of 2,3-butanediol production from glycerol and acid-pretreated rice straw hydrolysate by newly isolated strains: pre-evaluation as an integrated biorefinery process. Bioresour Technol 135:446–453. doi:10.1016/j.biortech.2012.10.141
Ji XJ, Huang H, Zhu JG, Ren LJ, Nie ZK, Du J, Li S (2010) Engineering Klebsiella oxytoca for efficient 2,3-butanediol production through insertional inactivation of acetaldehyde dehydrogenase gene. Appl Microbiol Biotechnol 85:1751–1758. doi:10.1007/s00253-009-2222-2
Ji XJ, Huang H, Ouyang PK (2011) Microbial 2,3-butanediol production: a state-of-the-art review. Biotechnol Adv 29:351–364. doi:10.1016/j.biotechadv.2011.01.007
Jung MY, Ng ChY, Song H, Lee J, Oh MK (2012) Deletion of lactate dehydrogenase in Enterobacter aerogenes to enhance 2,3-butanediol production. Appl Microbiol Biotechnol 95:461–469. doi:10.1007/s00253-012-3883-9
Jung MY, Park BS, Lee J, Oh MK (2013) Engineered Enterobacter aerogenes for efficient utilization of sugarcane molasses in 2,3-butanediol production. Bioresour Technol 139:21–27. doi:10.1016/j.biortech.2013.04.003
Jurchescu IM, Hamann J, Zhou X, Ortmann T, Kuenz A, Prüße U, Lang S (2013) Enhanced 2,3-butanediol production in fed-batch cultures of free and immobilized Bacillus licheniformis DSM 8785. Appl Microbiol Biotechnol 97:6715–6723. doi:10.1007/s00253-013-4981-z
Kallio PT, Bailey JE (1996) Intracellular expression of Vitreoscilla hemoglobin (VHb) enhances total protein secretion and improves the production of α-amylase and neutral protease in Bacillus subtilis. Biotechnol Prog 12:31–39. doi:10.1021/bp950065j
Krauze S (1962) Food analyst laboratory manual. PZWL, Warsaw
Laemmli UK (1970) Cleavage of structural protein during the assembly of the head of bacteriophage T4. Nature 227:680–685. doi:10.1038/227680a0
Li ZJ, Jian J, Wei XX, Shen XW, Chen GQ (2010) Microbial production of meso-2,3-butanediol by metabolically engineered Escherichia coli under low oxygen conditions. Appl Microbiol Biotechnol 87:2001–2009. doi:10.1007/s00253-010-2676-2
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:420–428. doi:10.1021/ac60147a030
Moes J, Griot M, Keller J, Heinzle E, Dunn IJ, Bourne JR (1985) A microbial culture with oxygen-sensitive product distribution as a potential tool for characterizing bioreactor oxygen transport. Biotechnol Bioeng 27:482–487. doi:10.1002/bit.260270413
Nawirska A, Kwaśniewska M (2005) Dietary fiber fractions from fruit and vegetable processing waste. Food Chem 91:221–225. doi:10.1016/j.foodchem.2003.10.005
Nielsen DR, Yoon SH, Yuan CJ, Prather LJ (2010) Metabolic engineering of acetoin and meso-2,3-butanediol biosynthesis in E. coli. Biotechnol J 5:274–284. doi:10.1002/biot.200900279
Nilegaonkar S, Bhosale SB, Kshirsagar DC, Kapadi AH (1992) Production of 2,3-butanediol from glucose by Bacillus licheniformis. World J Microbiol Biotechnol 8:378–381. doi:10.1007/BF01198748
Pyć R, Antczak T, Bratkowska H (2011) A method of enzymatic preparation production. Polish Patent no. 209161
Ruohonen L, Aristidou A, Frey AD, Penttila M, Kallio PT (2006) Expression of Vitreoscilla hemoglobin improves the metabolism of xylose in recombinant yeast Saccharomyces cerevisiae under low oxygen conditions. Enzym Microb Technol 39:6–14. doi:10.1016/j.enzmictec.2005.06.024
Slutter A, Hames B, Ruiz R, Scarlata C, Slutter J, Templeton D, Crocker D (2008) Determination of structural carbohydrates and lignin in biomass. NREL/TP-510-42618
Sun LH, Wang XD, Dai JY, Xiu ZL (2009) Microbial production of 2,3-butanediol from Jerusalem artichoke tubers by Klebsiella pneumoniae. Appl Microbiol Biotechnol 82:847–852. doi:10.1007/s00253-008-1823-5
Vendruscolo F, Albuquerque PM, Streit F, Esposito E, Ninow JL (2008) Apple pomace: a versatile substrate for biotechnological applications. Crit Rev Biotechnol 28:1–12. doi:10.1080/07388550801913840
Vishwakarma S (2014) Bioconversion of whey to 2,3-butanediol using Klebsiella oxytoca NRRL-13-199. Indian J Biotechnol 13:236–240
Wang A, Wang Y, Jiang T, Li L, Ma C, Xu P (2010) Production of 2,3-butanediol from corncob molasses, a waste by-product in xylitol production. Appl Microbiol Biotechnol 87:965–970. doi:10.1007/s00253-010-2557-8
Wang A, Xu Y, Ma C, Gao Ch, Li L, Wang Y, Tao F, Xu P (2012) Efficient 2,3-butanediol production from cassava powder by a crop-biomass-utilizer, Enterobacter cloacae subsp. dissolvens SDM. PLoS One 7:e40442. doi:10.1371/journal.pone.0040442
Xue GP, Johnson JS, Dalrymple BP (1999) High osmolarity improves the electro-transformation efficiency of the Gram-positive bacteria Bacillus subtilis and Bacillus licheniformis. J Microbiol Methods 34:183–191. doi:10.1016/S0167-7012(98)00087-6
Yang T, Rao Z, Zhang X, Lin Q, Xia H, Xu Z, Yang S (2011) Production of 2,3-butanediol from glucose by GRAS microorganism Bacillus amyloliquefaciens. J Basic Microbiol 51:650–658. doi:10.1002/jobm.201100033
Yang T, Rao Z, Zhang X, Xu M, Xu Z, Yang ST (2013a) Improved production of 2,3-butanediol in Bacillus amyloliquefaciens by over-expression of glyceraldehyde-3-phosphate dehydrogenase and 2,3-butandiol dehydrogenase. PLoS One 8:e76149. doi:10.1371/journal.pone.0076149
Yang TW, Rao ZM, Zhang X, Xu MJ, Xu ZH, Yang ST (2013b) Fermentation of biodiesel-derived glycerol by Bacillus amyloliquefaciens: effects of co-substrates on 2,3-butanediol production. Appl Microbiol Biotechnol 97:7651–7658. doi:10.1007/s00253-013-5048-x
Zhang L, Li Y, Wang Z, Xia Y, Chen W, Tang K (2007) Recent developments and future prospects of Vitreoscilla hemoglobin application in metabolic engineering. Biotechnol Adv 25:123–136. doi:10.1016/j.biotechadv.2006.11.001
Acknowledgments
Presented results were obtained within the scope of an international ERA-IB project (ERA-NET IB/02/2009) with an acronym PUBB. Financial support was provided by the Polish National Center for Research and Development (NCBiR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
ESM 1
(PDF 233 kb)
Rights and permissions
About this article
Cite this article
Białkowska, A.M., Jędrzejczak-Krzepkowska, M., Gromek, E. et al. Effects of genetic modifications and fermentation conditions on 2,3-butanediol production by alkaliphilic Bacillus subtilis . Appl Microbiol Biotechnol 100, 2663–2676 (2016). https://doi.org/10.1007/s00253-015-7164-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-7164-2