Abstract
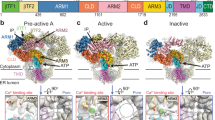
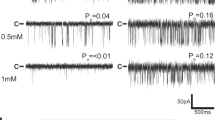
Inositol-1,4,5-trisphosphate receptors (InsP3Rs) and ryanodine receptors (RyRs) are tetrameric intracellular Ca2+ channels1. In each of these receptor families, the pore, which is formed by carboxy-terminal transmembrane domains, is regulated by signals that are detected by large cytosolic structures. InsP3R gating is initiated by InsP3 binding to the InsP3-binding core (IBC, residues 224–604 of InsP3R1)2 and it requires the suppressor domain (SD, residues 1–223 of InsP3R1)2,3,4,5,6,7,8. Here we present structures of the amino-terminal region (NT, residues 1–604) of rat InsP3R1 with (3.6 Å) and without (3.0 Å) InsP3 bound. The arrangement of the three NT domains, SD, IBC-β and IBC-α, identifies two discrete interfaces (α and β) between the IBC and SD. Similar interfaces occur between equivalent domains (A, B and C) in RyR1 (ref. 9). The orientations of the three domains when docked into a tetrameric structure of InsP3R10 and of the ABC domains docked into RyR9 are remarkably similar. The importance of the α-interface for activation of InsP3R and RyR is confirmed by mutagenesis and, for RyR, by disease-causing mutations9,11,12. Binding of InsP3 causes partial closure of the clam-like IBC, disrupting the β-interface and pulling the SD towards the IBC. This reorients an exposed SD loop (‘hotspot’ (HS) loop) that is essential for InsP3R activation7. The loop is conserved in RyR and includes mutations that are associated with malignant hyperthermia and central core disease9,11,12. The HS loop interacts with an adjacent NT, suggesting that activation re-arranges inter-subunit interactions. The A domain of RyR functionally replaced the SD in full-length InsP3R, and an InsP3R in which its C-terminal transmembrane region was replaced by that from RyR1 was gated by InsP3 and blocked by ryanodine. Activation mechanisms are conserved between InsP3R and RyR. Allosteric modulation of two similar domain interfaces within an N-terminal subunit reorients the first domain (SD or A domain), allowing it, through interactions of the second domain of an adjacent subunit (IBC-β or B domain), to gate the pore.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Serysheva, I., ed. Structure and Function of Calcium Release Channels. (Academic Press, 2010)
Bosanac, I. et al. Structure of the inositol 1,4,5-trisphosphate receptor binding core in complex with its ligand. Nature 420, 696–700 (2002)
Bosanac, I. et al. Crystal structure of the ligand binding suppressor domain of type 1 inositol 1,4,5-trisphosphate receptor. Mol. Cell 17, 193–203 (2005)
Rossi, A. M. et al. Synthetic partial agonists reveal key steps in IP3 receptor activation. Nature Chem. Biol. 5, 631–639 (2009)
Uchida, K., Miyauchi, H., Furuichi, T., Michikawa, T. & Mikoshiba, K. Critical regions for activation gating of the inositol 1,4,5-trisphosphate receptor. J. Biol. Chem. 278, 16551–16560 (2003)
Schug, Z. T. & Joseph, S. K. The role of the S4–S5 linker and C-terminal tail in inositol 1,4,5-trisphosphate receptor function. J. Biol. Chem. 281, 24431–24440 (2006)
Chan, J. et al. Structural studies of inositol 1,4,5-trisphosphate receptor: coupling ligand binding to channel gating. J. Biol. Chem. 285, 36092–36099 (2010)
Yamazaki, H., Chan, J., Ikura, M., Michikawa, T. & Mikoshiba, K. Tyr-167/Trp-168 in type1/3 inositol 1,4,5-trisphosphate receptor mediates functional coupling between ligand binding and channel opening. J. Biol. Chem. 285, 36081–36091 (2010)
Tung, C. C., Lobo, P. A., Kimlicka, L. & Van Petegem, F. The amino-terminal disease hotspot of ryanodine receptors forms a cytoplasmic vestibule. Nature 468, 585–588 (2010)
Ludtke, S. J. et al. Flexible architecture of IP3R1 by cryo-EM. Structure 19, 1192–1199 (2011)
Amador, F. J. et al. Crystal structure of type I ryanodine receptor amino-terminal β-trefoil domain reveals a disease-associated mutation “hot spot” loop. Proc. Natl Acad. Sci. USA 106, 11040–11044 (2009)
Lobo, P. A. & Van Petegem, F. Crystal structures of the N-terminal domains of cardiac and skeletal muscle ryanodine receptors: insights into disease mutations. Structure 17, 1505–1514 (2009)
Taylor, C. W. & Tovey, S. C. IP3 receptors: toward understanding their activation. Cold Spring Harb. Perspect. Biol. 2, a004010 (2010)
Anyatonwu, G. & Joseph, S. K. Surface accessibility and conformational changes in the N-terminal domain of type I inositol trisphosphate receptors: studies using cysteine substitution mutagenesis. J. Biol. Chem. 284, 8093–8102 (2009)
Tateishi, H. et al. Defective domain-domain interactions within the ryanodine receptor as a critical cause of diastolic Ca2+ leak in failing hearts. Cardiovasc. Res. 81, 536–545 (2009)
Sureshan, K. M. et al. Activation of IP3 receptors by synthetic bisphosphate ligands. Chem. Commun. 14, 1204–1206 (2009)
Chan, J. et al. Ligand-induced conformational changes via flexible linkers in the amino-terminal region of the inositol 1,4,5-trisphosphate receptor. J. Mol. Biol. 373, 1269–1280 (2007)
Mayer, M. L. Glutamate receptors at atomic resolution. Nature 440, 456–462 (2006)
Lin, C. C., Baek, K. & Lu, Z. Apo and InsP3-bound crystal structures of the ligand-binding domain of an InsP3 receptor. Nature Struct. Mol. Biol. 18, 1172–1174 (2011)
Hamada, T., Bannister, M. L. & Ikemoto, N. Peptide probe study of the role of interaction between the cytoplasmic and transmembrane domains of the ryanodine receptor in the channel regulation mechanism. Biochemistry 46, 4272–4279 (2007)
Ramos-Franco, J., Galvan, D., Mignery, G. A. & Fill, M. Location of the permeation pathway in the recombinant type-1 inositol 1,4,5-trisphosphate receptor. J. Gen. Physiol. 114, 243–250 (1999)
Samsó, M., Feng, W., Pessah, I. N. & Allen, P. D. Coordinated movement of cytoplasmic and transmembrane domains of RyR1 upon gating. PLoS Biol. 7, e85 (2009)
Iwai, M., Michikawa, T., Bosanac, I., Ikura, M. & Mikoshiba, K. Molecular basis of the isoform-specific ligand-binding affinity of inositol 1,4,5-trisphosphate receptors. J. Biol. Chem. 282, 12755–12764 (2007)
Chu, A., Diaz-Munoz, M., Hawkes, M. J., Brush, K. & Hamilton, S. L. Ryanodine as a probe for the functional state of the skeletal muscle sarcoplasmic reticulum calcium release channel. Mol. Pharmacol. 37, 735–741 (1990)
Lai, F. A. & Meissner, G. The muscle ryanodine receptor and its intrinsic Ca2+ channel activity. J. Bioenerg. Biomembr. 21, 227–246 (1989)
Liu, Z. et al. Dynamic, inter-subunit interactions between the N-terminal and central mutation regions of cardiac ryanodine receptor. J. Cell Sci. 123, 1775–1784 (2010)
George, C. H. et al. Ryanodine receptor regulation by intramolecular interactions between cytoplasmic and transmembrane domains. Mol. Biol. Cell 15, 2627–2638 (2004)
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007)
Otsu, K. et al. Molecular cloning of cDNA encoding the Ca2+ release channel (ryanodine receptor) of rabbit cardiac muscle sarcoplasmic reticulum. J. Biol. Chem. 265, 13472–13483 (1990)
Tovey, S. C. et al. Regulation of inositol 1,4,5-trisphosphate receptors by cAMP independent of cAMP-dependent protein kinase. J. Biol. Chem. 285, 12979–12989 (2010)
Kenakin, T. P. Pharmacologic Analysis of Drug–Receptor Interactions 3rd edn (Lippincott, Williams & Wilkins, 1997)
Murshudov, G. N., Vagin, A. A. & Dodson, E. J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D 53, 240–255 (1997)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004)
Zorzato, F. et al. Molecular cloning of cDNA encoding human and rabbit forms of the Ca2+ release channel (ryanodine receptor) of skeletal muscle sarcoplasmic reticulum. J. Biol. Chem. 265, 2244–2256 (1990)
Tovey, S. C., Sun, Y. & Taylor, C. W. Rapid functional assays of intracellular Ca2+ channels. Nature Protocols 1, 259–263 (2006)
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004)
Wriggers, W., Milligan, R. A. & McCammon, J. A. Situs: a package for docking crystal structures into low-resolution maps from electron microscopy. J. Struct. Biol. 125, 185–195 (1999)
Acknowledgements
We thank P. Allen and D. MacLennan for gifts of plasmids encoding RyR2 and RyR1, respectively. C.W.T. thanks T. Rahman and V. Konieczny for discussions. M.I. acknowledges K. Mikoshiba and T. Michikawa for long-standing support and discussions. This work was supported by grants from the Heart and Stroke Foundation of Ontario (T-7181) to M.I., National Institutes of Health Research (EY012347 and NS059969) to J.B.A., the Wellcome Trust (085295), the Biotechnology and Biological Sciences Research Council (BB/H009736) and the Medical Research Council (G0900049) to C.W.T. M.-D.S. is supported by postdoctoral fellowships from the Canadian Institutes of Health Research and the National Research Foundation of Korea (2009-352-E00006). A.M.R. is a fellow of Queens’ College, Cambridge. M.I. holds a Canadian Research Chair in Cancer Structural Biology.
Author information
Authors and Affiliations
Contributions
Author Contributions M.-D.S., N.I., P.B.S., M.I. and C.L. determined and analysed the structure of NT. S.V. prepared and characterized the full-length InsP3R and chimaeras. A.M.R., S.A.K. and P.D. completed analyses of InsP3 binding and related molecular biology. J.B.A., M.I. and C.W.T. supervised work in their respective laboratories, coordinated the project and, with input from other authors, wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains a Supplementary Discussion, Supplementary References, Supplementary Tables 1-9 and Supplementary Figures 1-11 with legends. (PDF 3054 kb)
Rights and permissions
About this article
Cite this article
Seo, MD., Velamakanni, S., Ishiyama, N. et al. Structural and functional conservation of key domains in InsP3 and ryanodine receptors. Nature 483, 108–112 (2012). https://doi.org/10.1038/nature10751
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature10751
This article is cited by
-
Structural titration reveals Ca2+-dependent conformational landscape of the IP3 receptor
Nature Communications (2023)
-
Ligand sensitivity of type-1 inositol 1,4,5-trisphosphate receptor is enhanced by the D2594K mutation
Pflügers Archiv - European Journal of Physiology (2023)
-
RyR1-related myopathy mutations in ATP and calcium binding sites impair channel regulation
Acta Neuropathologica Communications (2021)
-
Retrieving functional pathways of biomolecules from single-particle snapshots
Nature Communications (2020)
-
Second messenger Ap4A polymerizes target protein HINT1 to transduce signals in FcεRI-activated mast cells
Nature Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.