Abstract
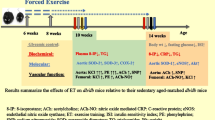
Superoxide (O ·−2 ) overproduction, by decreasing the nitric oxide (·NO) bioavailability, contributes to vascular complications in type 1 diabetes. In this disease, the vascular O ·−2 can be produced by the NADPH oxidase (NOX), nitric oxide synthase (NOS), and xanthine oxidase (XO). This study aimed to determine the contribution of each enzymatic pathway in hyperglycemia-induced O ·−2 overproduction, and the effects of an endurance training program and insulin therapy, associated or not, on the O ·−2 production (amount and related enzymes) in diabetic rats. Forty male Wistar rats were divided into diabetic (D), diabetic treated with insulin (D-Ins), diabetic trained (D-Tr), or diabetic insulin-treated and trained (D-Ins + Tr) groups. An additional healthy group was used as control. Insulin therapy (Glargine Lantus, Sanofi) and endurance training (treadmill run: 60 min/day, 25 m/min, 5 days/week) started 1 week after diabetes induction by streptozotocin (45 mg/kg), and lasted for 8 weeks. At the end of the protocol, the O ·−2 production in aorta rings was evaluated by histochemical analyses (DHE staining). Each production pathway was studied by inhibiting NOX (apocynin), NOS (L-Name), or XO (allopurinol) before DHE staining. Diabetic rats exhibited hyperglycemia-induced O ·−2 overproduction, resulting from NOX, NOS, and XO activation. Insulin therapy and endurance training, associated or not, decreased efficiently and similarly the O ·−2 overproduction. Insulin therapy reduced the hyperglycemia and decreased the three enzymatic pathways implicated in the O ·−2 production. Endurance training decreased directly the NOS and XO activity. While both therapeutic strategies activated different pathways, their association did not reduce the O ·−2 overproduction more significantly.



Similar content being viewed by others
Notes
Among the two types of NOS isoforms in aorta, uncoupled eNOS has been reported as the major source of superoxide anion production, comparative to iNOS (negligible). Hereinafter, L-Name was considered to essentially inhibit superoxide anion production by eNOS uncoupling.
References
De Vriese AS, Verbeuren TJ, Van de Voorde J, Lameire NH, Vanhoutte PM (2000) Endothelial dysfunction in diabetes. Br J Pharmacol 130:963–974
Pacher P, Beckman JS, Liaudet L (2007) Nitric oxide and peroxynitrite in health and disease. Physiol Rev 87:315–424
Chang KC, Chung SY, Chong WS, Suh JS, Kim SH, Noh HK et al (1993) Possible superoxide radical-induced alteration of vascular reactivity in aortas from streptozotocin-treated rats. J Pharmacol Exp Ther 266:992–1000
Guzik TJ, West NE, Black E, McDonald D, Ratnatunga C, Pillai R et al (2000) Vascular superoxide production by NAD(P)H oxidase: association with endothelial dysfunction and clinical risk factors. Circ Res 86:E85–E90
Liu S, Ma X, Gong M, Shi L, Lincoln T, Wang S (2007) Glucose down-regulation of cGMP-dependent protein kinase I expression in vascular smooth muscle cells involves NAD(P)H oxidase-derived reactive oxygen species. Free Radic Biol Med 42:852–863
Guzik TJ, Mussa S, Gastaldi D, Sadowski J, Ratnatunga C, Pillai R et al (2002) Mechanisms of increased vascular superoxide production in human diabetes mellitus: role of NAD(P)H oxidase and endothelial nitric oxide synthase. Circulation 105:1656–1662
Dikalova AE, Gongora MC, Harrison DG, Lambeth JD, Dikalov S, Griendling KK (2010) Upregulation of Nox1 in vascular smooth muscle leads to impaired endothelium-dependent relaxation via eNOS uncoupling. Am J Physiol Heart Circ Physiol 299:H673–H679
Matsumoto S, Koshiishi I, Inoguchi T, Nawata H, Utsumi H (2003) Confirmation of superoxide generation via xanthine oxidase in streptozotocin-induced diabetic mice. Free Radic Res 37:767–772
Houston M, Estevez A, Chumley P, Aslan M, Marklund S, Parks DA et al (1999) Binding of xanthine oxidase to vascular endothelium. Kinetic characterization and oxidative impairment of nitric oxide-dependent signaling. J Biol Chem 274:4985–4994
Wiernsperger NF (2003) Oxidative stress as a therapeutic target in diabetes: revisiting the controversy. Diabetes Metab 29:579–585
Karasu C, Altan VM (1993) The role of endothelial cells on the alterations in vascular reactivity induced by insulin-dependent diabetes mellitus: effects of insulin treatment. Gen Pharmacol 24:743–755
Adams V, Linke A, Krankel N, Erbs S, Gielen S, Mobius-Winkler S et al (2005) Impact of regular physical activity on the NAD(P)H oxidase and angiotensin receptor system in patients with coronary artery disease. Circulation 111:555–562
Moien-Afshari F, Ghosh S, Elmi S, Rahman MM, Sallam N, Khazaei M et al (2008) Exercise restores endothelial function independently of weight loss or hyperglycaemic status in db/db mice. Diabetologia 51:1327–1337
Szkudelski T (2001) The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiol Res 50:537–546
Le Douairon LS, Rebillard A, Zguira MS, Malarde L, Saiag B, Gratas-Delamarche A et al (2011) Effects of exercise training combined with insulin treatment on cardiac NOS1 signaling pathways in type 1 diabetic rats. Mol Cell Biochem 347:53–62
Brownlee M (2005) The pathobiology of diabetic complications: a unifying mechanism. Diabetes 54:1615–1625
Peterson TE, Poppa V, Ueba H, Wu A, Yan C, Berk BC (1999) Opposing effects of reactive oxygen species and cholesterol on endothelial nitric oxide synthase and endothelial cell caveolae. Circ Res 85:29–37
Suh YA, Arnold RS, Lassegue B, Shi J, Xu X, Sorescu D et al (1999) Cell transformation by the superoxide-generating oxidase Mox1. Nature 401:79–82
Karasu C (2010) Glycoxidative stress and cardiovascular complications in experimentally-induced diabetes: effects of antioxidant treatment. Open Cardiovasc Med J 4:240–256
Shen GX (2010) Oxidative stress and diabetic cardiovascular disorders: roles of mitochondria and NADPH oxidase. Can J Physiol Pharmacol 88:241–248
Kakehi T, Yabe-Nishimura C (2008) NOX enzymes and diabetic complications. Semin Immunopathol 30:301–314
Lambertucci RH, Levada-Pires AC, Rossoni LV, Curi R, Pithon-Curi TC (2007) Effects of aerobic exercise training on antioxidant enzyme activities and mRNA levels in soleus muscle from young and aged rats. Mech Ageing Dev 128:267–275
Rush JW, Denniss SG, Graham DA (2005) Vascular nitric oxide and oxidative stress: determinants of endothelial adaptations to cardiovascular disease and to physical activity. Can J Appl Physiol 30:442–474
Acknowledgments
The authors thank the platform of fluorescence microscopy (H2P2, Rennes), Pascale Bellaud and Roselyne Viel for their technical support.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Malardé, L., Rebillard, A., Le Douairon-Lahaye, S. et al. Superoxide production pathways in aortas of diabetic rats: beneficial effects of insulin therapy and endurance training. Mol Cell Biochem 389, 113–118 (2014). https://doi.org/10.1007/s11010-013-1932-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1932-z