Abstact
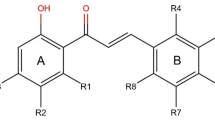
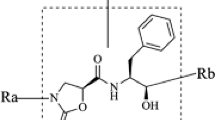
3D-QSAR studies using the Comparative Molecular Field Analysis (CoMFA) methodology were conducted to predict the inhibition constants, Ki, and the inhibitor concentrations, IC90 of 127 symmetrical and unsymmetrical cyclic urea and cyclic cyanoguanidine derivatives containing different substituent groups such as: benzyl, isopropyl, 4-hydroxybenzyl, ketone, oxime, pyrazole, imidazole, triazole and having anti-HIV-1 protease activities. A significant cross-validated correlation coefficient (q2) of 0.63 and a fitted correlation coefficient r2 of 0.70 were obtained, indicating that the models can predict the anti-protease activity from poorly to highly active compounds reliably. The best predictions were obtained for: XV643 (predicted log 1/Ki=9.86), a 3,5-dimethoxy-benzyl cyclic urea derivate (molec60, predicted log 1/Ki=8.57) and a benzyl cyclic urea derivate (molec 61, predicted log 1/IC90=6.87). Using the CoMFA method, we also predicted the biological activity of 14 cyclic urea derivatives that inhibit the HIV-1 protease mutants V82A, V82I and V82F. The predicted biological activities of the: (i) XNO63 (inhibitory activity on the mutant HIV-1 PR V82A), (ii) SB570 (inhibiting the mutant HIV-1 PR V82I) and also (iii) XV652 (during the interaction with the mutant HIV-1 PR V82F) were in good agreement with the experimental values.
Figure Stereoview of the contour plots of the CoMFA steric and electrostatic fields. The favorable (indicated by blue polyhedra) and unfavorable (represented by red polyhedra) electrostatic areas and also the favorable (shown by green polyhedra) and unfavorable (shown by yellow polyhedra) steric areas formed around the most active molecule, 6a.



Similar content being viewed by others
References
Wu J, Adomat MR, Tleis MJ, Leis J, Harrison RW, Weber I (1998) Biochemistry 37:4518–4526
Klabe RM, Bacheler LT, Ala PJ (1998) Biochemistry 37:8735–8742
Louis J, Wondrack ME, Kimmel RA, Wingfield T, Nashed TN (1999) J Biol Chem 274:23437–23442
Wlodawer A, Vondrasek J (1998) Annu Rev Biophys Struct 27:249–284
Vega S, Kang LW, Velazquez-Campoy A, Kiso Y, Amzel LM, Freire E (2004) Proteins 55:594–602
Konings FAJ, Zhong P, Agwara M, Agyingi L, Zekeng L, Achkar JM, Ewane L, Saa Z, Emmanuel A, Kinge T, Nyambi PN (2004) AIDS Res Hum Retroviruses 20:105–109
Oprea TI, Waller LC (1997) J Comp Chem 11:127–12
Nugiel A, Seitz J (1996) J Med Chem 39:2156–2169
Jadkav PK, Woerner FJ, Lam PYS, Hodge CN, Eyermnn CJ, Man HW, Daneker WF, Bacheler LT, Rayner MM, Meek JL, Erickson-Viitanen S, Jackson DA, Calabrese JC, Schadt M, Chong-Hwan C (1998) J Med Chem 41:1446–1455
Lin TH, Li HT, Tsai KC (2004) J Chem Inf Comput Sci 44:76–87
Senese CL, Hopfinger AJ (2003) J Chem Inf Comput Sci 43:2180–2193
Senese CL, Hopfinger AJ (2003) J Chem Inf Comput Sci 43:1297–1307
Kurup A, Mekapati SB, Garg R, Hansch C (2003) Curr Med Chem 10:1679–1688
Kiralj R, Ferreira MM (2003) J Mol Graph Model 21:435–448
Waller LC, Oprea TI, Giolitti A, Marshall RG (1993) J Med Chem 36:4152–4160
Schaal W, Karlsson A, Ahlsen G, Lindberg J, Andersson HO, Danielson UH, Classon B, Unge T, Samuelsson B, Hulten J, Hallberg A, Karlen A (2001) J Med Chem 44:155–169
Buolamwini JK, Assefa HJ (2002) J Med Chem 45:841–852
Nair AC, Jayatilleke P, Wang X, Miertus S, Welsh W (2002) J Med Chem 45:973–983
Huang X, Xu L, Luo X, Fan K, Ji R, Pei G, Chen K, Jiang H (2002) J Med Chem 45:333–343
Avram S, Bologa C, Banda M, Flonta ML (2001) Romanian J Biophys 11:11–24
Avram S, Svab I, Bologa C, Flonta ML (2003) J Cell Mol Med 7:287–296
Svab I, Avram S, Flonta ML (2002) Romanian J Biophys 11:119–132
Avram S, Movileanu L, Mihailescu D, Flonta ML (2002) J Cell Mol Med 6:251–260
Han Q, Chang CH, Li R, Ru Y, Jadhav PK, Lam PYS (1998) J Med Chem 41:2019–2028
Lam PY, Ru Y, Jadhav PK, Aldrich PE, DeLucca GV, Eyermann CJ, Chang CH, Emmett G, Holler ER, Daneker WF, Li L, Confalone PN, McHugh RJ, Han Q, Li R, Markwalder JA, Seitz SP, Sharpe TR, Bacheler LT, Rayner MM, Klabe RM, Shum L, Winslow DL, Kornhauser DM, Hodge CN (1996) J Med Chem 30:3514–3525
Hylton K-G, Main AD, McElwee-White L (2003) J Org Chem 68:1615–1617
Golub G, O’leary D (1989) SIAM Rev 31:50–102
Sybyl Theory Manual (1988) Tripos Associates Inc., 1699 South Hanley Road, St. Louis, MO 63144
Sybyl Molecular Spreadsheet Manual (1988) Tripos Associates Inc., 1699 South Hanley Road, St. Louis, MO 63144
Sybyl Tutorial Manual (1988) Tripos Associates Inc., 1699 South Hanley Road, St. Louis, MO 63144
Sybyl Basic Ligand-Based Design Manual (1988) Tripos Associates Inc., 1699 South Hanley Road, St. Louis, MO 63144
Cho JS, Tropsha A (1995) J Med Chem 38:1060–1066
Acknowledgements
We thank Dr. Andrei Petrescu, Biochemistry Institute, Bucharest, for fruitful discussion.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Avram, S., Bologa, C. & Flonta, ML. Quantitative structure-activity relationship by CoMFA for cyclic urea and nonpeptide-cyclic cyanoguanidine derivatives on wild type and mutant HIV-1 protease. J Mol Model 11, 105–115 (2005). https://doi.org/10.1007/s00894-004-0226-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-004-0226-5