Abstract
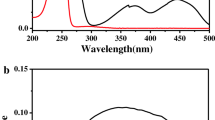
Pronase treatment of aqueous suspensions of purple membrane fragments from H. halobium leads to the cleavage of bacteriorhodopsin. The protein fragments remaining in the membrane after treatment with relatively small concentrations of enzyme (2% w/w) in normal daylight range in molecular weight from 20,000-21,000 daltons, indicating that cleavage occurs mainly near the extremities of the protein chain. At higher enzyme concentrations the relative amounts of protein fragments having smaller molecular weight increase. Generally, the relative loss of retinal chromophore is larger than that of protein and thus the retinal binding site seems to be located near one of the chain ends that is cleaved off by enzyme.
Irradiation with white light during the time of proteolysis (at both low and high enzyme concentrations) results in extensive cleavage, so that under certain conditions no high molecular weight components can be detected in SDS-polyacrylamide gels. It, therefore, appears that parts of the bacteriorhodopsin chain become more exposed to enzyme digestion when the purple membrane is illuminated.
Enzyme treated aqueous purple membrane fragment suspensions still show photocycle activity. The main consequence of proteolysis is a pronounced appearance of biphasicity in the decay of M412 and the regeneration of bR570. Simultaneously the yield of O660 is reduced. As with untreated purple membrane, the correlation between the rates of decay of M412 and regeneration of bR570 is greatest when the yield of O660 is lowest.
Similar content being viewed by others
References
Bridgen, J., Walker, I. D.: Photoreceptor protein from the purple membrane of Halobacterium halobium: Molecular weight and retinal binding site. Biochemistry 15, 792–798 (1976)
Brith-Lindner, M., Rosenheck, K.: The circular dichroism of bacteriorhodopsin: Asymmetry and light-scattering distortions. FEBS-Lett. 76, 41–44 (1977)
Caplan, S. R., Eisenbach, E., Cooper, S., Garty, H., Klemperer, G., Bakker, E. P.: Light-driven proton and sodium ion transport in bacteriorhodopsin-containing particles. In: Bioenergetics of Membranes (eds. L. Packer et al.), pp. 101–114. Amsterdam: Elsevier/North Holland Biomedical Press 1977
Chou, P. Y., Fasman, G. D.: Prediction of protein conformation. Biochemistry 13, 222–245 (1974)
Chukung, M., Devault, D., Hess, B., Oesterhelt, D.: Photolysis of bacteriorhodopsin. Biophys. J. 15, 907–911 (1975)
Danon, A., Stoeckenius, W.: Photophosphorylation in Halobacterium halobium. Proc. nat. Acad. Sci. (Wash.) 71, 1234–1238 (1974)
Dencher, N., Wilms, M.: Flash photometric experiments on the photochemical cycle of bacteriorhodopsin. Biophys. Struct. Mechanism 1, 259–271 (1975)
Eisenbach, M., Bakker, E. P., Korenstein, R., Caplan, S. R.: Bacteriorhodopsin: Biphasic kinetics of phototransients and of light-induced proton transfer by sub-bacterial H. halobium particles and by reconstituted liposomes. FEBS-Lett. 71, 228–232 (1976)
Eisenbach, M., Garty, H., Klemperer, G., Weissmann, C., Tanny, G., Caplan, S. R.: Light-induced pH changes in purple-membrane fragments of Halobacterium halobium. In: Bioenergetics of Membranes (eds. L. Packer et al.), pp. 119–128. Amsterdam: Elsevier/North Holland Biomedical Press 1977
Fisher, E.: Simple cooling arrangements for absorption, emission and ESR spectrophotometry. Mol. Photochem. 2, 99–101 (1970)
Garty, H., Klemperer, G., Eisenbach, M., Caplan, S. R.: The direction of light-induced pH changes in purple membrane suspensions: Influence of pH and temperature. FEBS-Lett. 81, 238–242 (1977)
Gerber, G. E., Wildenauer, D., Khorana, H. G.: Proteolytic studies of the asymmetric orientation of bacteriorhodopsin in synthetic lipid vesicles. Fed. Proc. Abstr. of 61 Ann. Meeting, p. 896 (1977)
Henderson, R., Unwin, P. N. T.: Three-dimensional model of purple membrane obtained by electron microscopy. Nature (Lond.) 257, 28–32 (1975)
Hess, B., Kuschmitz, D.: The photochemical reaction of the 412 nm chromophore of bacteriorhodopsin. FEBS-Lett. 74, 20–24 (1977)
Kaufmann, K. J., Rentzepies, P. M., Stoeckenius, W., Lewis, A.: Primary photochemical processes in bacteriorhodopsin. Biochem. biophys. Res. Commun. 68, 1109–1115 (1976)
Lewis, A., Spoonhower, J., Bogomolni, R. A., Lozier, R. H., Stoeckenius, W.: Tunable laser raman spectroscopy of bacteriorhodopsin. Proc. nat. Acad. Sci. (Wash.) 71, 4462–4466 (1974)
Lewis, P. N., Momany, F. A., Scheraga, H. A.: Folding of polypeptide chains in proteins: A proposed mechanism for folding. Proc. nat. Acad. Sci. (Wash.) 68, 2293–2297 (1971)
Lowry, O. H., Rosebrough, N. J., Farr, A. L., Randall, R. J.: Protein measurement with the folin phenol reagent. J. biol. Chem. 193, 265–275 (1951)
Lozier, R. H., Bogomolni, R. A., Stoeckenius, W.: Bacteriorhodopsin: A light-driven proton pump in H. halobium. Biophys. J. 15, 955–962 (1975)
Oesterhelt, D., Stoeckenius, W.: Rhodopsin-like protein from the purple membrane of Halobacterium halobium. Nature (Lond.) New Biol. 233, 149–152 (1971)
Oesterhelt, D., Stoeckenius, W.: Functions of a new photoreceptor membrane. Proc. nat. Acad. Sci. (Wash.) 70, 2853–2857 (1973)
Oesterhelt, D., Schuhmann, L., Gruber, H.: Light-dependent reaction of bacteriorhodopsin with hydroxylamine in cell suspensions of Halobacterium halobium: Demonstration of an apo-membrane. FEBS-Lett. 44, 257–261 (1974)
Peters, J., Peters, R., Stoeckenius, W.: A photosensitive product of sodium borohydride reduction of bacteriorhodopsin. FEBS-Lett. 61, 128–134 (1976)
Sherman, W. V., Slifkin, M. A., Caplan, S. R.: Kinetic studies of phototransients in bacteriorhodopsin. Biochim. biophys. Acta (Amst.) 423, 238–248 (1976a)
Sherman, W. V., Korenstein, R., Caplan, S. R.: Energetics and chronology of phototransients in the light response of the purple membrane of H. halobium. Biochim. biophys. Acta (Amst.) 430, 454–458 (1976b)
Slifkin, M. A., Caplan, S. R.: Modulation excitation spectrophotometry of purple membrane of Halobacterium halobium. Nature (Lond.) 253, 56–58 (1975)
Venkatachalam, C. M.: Stereochemical criteria for polypeptides and proteins: V. Conformation of a system of three linked peptide units. Biopolymers 6, 1425–1436 (1968)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rosenheck, K., Brith-Lindner, M., Lindner, P. et al. Proteolysis and flash photolysis of bacteriorhodopsin in purple membrane fragments. Biophys. Struct. Mechanism 4, 301–313 (1978). https://doi.org/10.1007/BF00537613
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00537613