Abstract
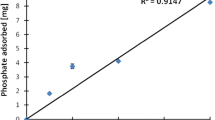
Lanthanum-modified bentonite has potential for wide application in eutrophication control. We investigated P adsorption on a lanthanum-modified bentonite by analysis of adsorption kinetics, equilibrium, and the effect of environmental factors. P adsorption closely followed the pseudo-second-order kinetic model, and the isotherm was well described by the Langmuir model. This adsorbent could effectively immobilize P into the sediment, but the adsorption process was strongly dependent on pH, anions, and low molecular weight organic acids (LMWOAs). P adsorption increased with increasing pH from 0.52 mg P/g at pH 3.0 to 0.93 mg P/g at pH 7.0 with no adsorption at pH 11. P adsorption was strongly inhibited in the presence of anions and three LMWOAs, with P even re-released at high concentrations. These environmental factors should be given significant attention when considering the application of lanthanum-modified bentonite in eutrophication control.






Similar content being viewed by others
References
Albert DB, Taylor C, Martens CS (1995) Sulfate reduction rates and low molecular weight fatty acid concentations in the water column and surficial sediments of the Black Sea. Deep Res I 42:1239–1260
Ao H, Wu C, Xiong X, Jing L, Huang X, Zhang K, Liu J (2014) Water and sediment quality in Qinghai Lake, China: a revisit after half a century. Environ Monit Assess 186:2121–2133
APHA (1999) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association, Washington
Berkowitz J, Anderson MA, Amrhein C (2006) Influence of aging on phosphorus sorption to alum floc in lake water. Water Res 40:911–916
Boström B, Persson G, Broberg B (1988) Bioavailability of different phosphorus forms in freshwater systems. Hydrobiologia 170:133–155
Chen N, Feng C, Zhang Z, Liu R, Gao Y, Li M, Sugiura N (2012) Preparation and characterization of lanthanum(III) loaded granular ceramic for phosphorus adsorption from aqueous solution. J Taiwan Inst Chem Eng 43:783–789
Freundlich H (1906) Concerning adsorption in solutions. Z Phys Chem 57:385–470
Ho YS, McKay G (1999) The sorption of lead(II) ions on peat. Water Res 33:578–584
Ho YS, McKay G (2000) The kinetics of sorption of divalent metal ions onto sphagnum moss peat. Water Res 34:735–742
Huang W, Chen J, He F, Tang J, Li D, Zhu Y, Zhang Y (2015) Effective phosphate adsorption by Zr/Al-pillared montmorillonite: insight into equilibrium, kinetics and thermodynamics. Appl Clay Sci 104:252–260
Huser BJ (2012) Variability in phosphorus binding by aluminum in alum treated lakes explained by lake morphology and aluminum dose. Water Res 46:4697–4704
Jeppesen E, Søndergaard M, Jensen JP, Havens KE, Anneville O, Carvalho L, Coveney MF, Deneke R, Dokulil MT, Foy B, Gerdeaux D, Hampton SE, Hilt S, Kangur K, Kohler J, Lammens EH, Lauridsen TL, Manca M, Miracle MR, Moss B, Noges P, Persson G, Phillips G, Portielje R, Romo S, Schelske CL, Straile D, Tatrai I, Willen E, Winder M (2005) Lake responses to reduced nutrient loading—an analysis of contemporary long-term data from 35 case studies. Freshw Biol 50:1747–1771
Jones EV (2004) Phosphorus in environmental technologies: principles and applications. IWA Publishing, London
Jones DLL, Dennis PGG, Owen AGG, van Hees PAW (2003) Organic acid behavior in soils—misconceptions and knowledge gaps. Plant Soil 248:31–41
Kuroki V, Bosco GE, Fadini PS, Mozetob A, Cestaric AR, Carvalhoa WA (2014) Use of a La(III)-modified bentonite for effective phosphate removal from aqueous media. J Hazard Mater 274:124–131
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica and platinum. J Am Chem Soc 40:1361–1403
Lewandowski J, Schauser I, Hupfer M (2003) Long-term effects of phosphorus precipitations with alum in hypereutrophic Lake Süsser See (Germany). Water Res 37:3194–3204
Li B, Zhang T (2013) Removal mechanisms and kinetics of trace tetracycline by two types of activated sludge treating freshwater sewage and saline sewage. Environ Sci Pollut Res 20:3024–3033
Liu G, Ye C, He J, Qian Q, Jiang H (2009) Lake sediment treatment with aluminum, iron, calcium and nitrate additives to reduce phosphorus release. J Zhejiang Univ Sci A 10:1367–1373
Liu J, Wan L, Zhang L, Zhou Q (2011) Effect of pH, ionic strength, and temperature on the phosphate adsorption onto lanthanum-doped activated carbon fiber. J Colloid Interface Sci 364:490–496
Lürling M, Van Oosterhout F (2013a) Controlling eutrophication by combined bloom precipitation and sediment phosphorus inactivation. Water Res 47:6527–6537
Lürling M, van Oosterhout F (2013b) Case study on the efficacy of a lanthanum-enriched clay (Phoslock®) in controlling eutrophication in Lake Het Groene Eiland (The Netherlands). Hydrobiologia 710:253–263
Lürling M, Waajen G, Van Oosterhout F (2014) Humic substances interfere with phosphate removal by lanthanum modified clay in controlling eutrophication. Water Res 54:78–88
Ma J, Yang K, Zhang XF, Li J (2013) Biological floating island technology for water remediation based on micro-nano bubble aeration methods. China Water Res 2:66–67
Márquez-Pacheco H, Hansen AM, Falcón-Rojas A (2013) Phosphorous control in a eutrophied reservoir. Environ Sci Pollut Res 20:8446–8456
Mehner T, Diekmann M, Gonsiorczyk T, Kasprzak P, Koschel R, Krienitz L, Rumpf M, Schulz M, Wauer G (2008) Rapid recovery from eutrophication of a stratified lake by disruption of internal nutrient load. Ecosystems 11:1142–1156
Paerl HW, Xu H, McCarthy MJ, Zhu G, Qin B, Li Y, Gardner WS (2011) Controlling harmful cyanobacterial blooms in a hyper-eutrophic lake (Lake Taihu, China): the need for a dual nutrient (N & P) management strategy. Water Res 45:1973–1983
Perkins RG, Underwood GJC (2001) The potential for phosphorus release across the sediment-water interface in an eutrophic reservoir dosed with ferric sulphate. Water Res 35:1399–1406
Qian WM, Yang H, Mei XY, Zhou SL (2008) Research on preparation of bentonite absorbent modified by La or La2Ti and its phosphor removal efficiency. Appl Chem Industry 37:54–66 (In Chinese)
Reitzel K, Jensen HS, Egemose S (2013) PH dependent dissolution of sediment aluminum in six Danish lakes treated with aluminum. Water Res 47:1409–1420
Ren YX, Nakano K, Nomura M, Chiba N, Nishimura O (2007) A thermodynamic analysis on adsorption of estrogens in activated sludge process. Water Res 41:2341–2348
Robb M, Greenop B, Goss Z, Douglas G, Adeney J (2003) Application of PhoslockTM, an innovative phosphorus binding clay, to two Western Australian waterways: Preliminary findings. Hydrobiologia 494:237–243
Ross G, Haghseresht F, Cloete TE (2008) The effect of pH and anoxia on the performance of Phoslock, a phosphorus binding clay. Harmful Algae 7:545–550
Ruttenberg KC (1992) Development of a sequential extraction method for different forms of phosphorus in marine sediments. Limnol Oceanogr 37:1460–1482
Shinneman A, Edlund MB, Almendinger JE, Soninkhishig N (2009) Diatoms as indicators of water quality in Western Mongolian lakes: a 54-site calibration set. J Paleolimnol 42:373–389
Søndergaard M, Jensen JP, Jeppesen E (2003) Role of sediment and internal loading of phosphorus in shallow lakes. Hydrobiologia 506–509:135–145
Spears BM, Meis S, Anderson A, Kellou M (2013) Comparison of phosphorus (P) removal properties of materials proposed for the control of sediment p release in UK lakes. Sci Total Environ 442:103–110
Tanada S, Kabayama M, Kawasaki N, Sakiyama T, Nakamura T, Araki M, Tamura T (2003) Removal of phosphate by aluminum oxide hydroxide. J Colloid Interface Sci 257:135–140
Wang Z, Nie E, Li J, Yang M, Zhao Y, Luo X, Zheng Z (2012) Equilibrium and kinetics of adsorption of phosphate onto iron-doped activated carbon. Environ Sci Pollut Res 19:2908–2917
Whitmore TJ, Brenner M, Jiang Z, Curtis JH, Moore AM, Engstrom DR, Wu Y (1997) Water quality and sediment geochemistry in lakes of Yunnan Province, southern China. Environ Geol 32:45–55
Wu J, Zeng H, Yu H, Ma L, Xu L, Qin B (2012) Water and sediment quality in lakes along the middle and lower reaches of the Yangtze River, China. Water Resour Manag 26:3601–3618
Xiao M, Wu FC, Liao HQ (2009) Vertical profiles of low molecular weight organic acids in sediment porewaters of six Chinese lakes. J Hydrol 365:37–45
Xiao M, Wu F, Liao H (2010) Characteristics and distribution of low molecular weight organic acids in the sediment porewaters in Bosten Lake, China. J Environ Sci 22:328–337
Xie J, Wang Z, Lu S, Wu D, Zhang Z, Kong H (2014) Removal and recovery of phosphate from water by lanthanum hydroxide materials. Chem Eng J 254:163–170
Yang M, Lin J, Zhan Y, Zhu Z, Zhang H (2014) Immobilization of phosphorus from water and sediment using zirconium-modified zeolites. Environ Sci Pollut Res 22:3606–3619
Zamparas M, Zacharias I (2014) Restoration of eutrophic freshwater by managing internal nutrient loads. A review. Sci Total Environ 496:551–562
Zamparas M, Gianni A, Stathi P, Deligiannakis P, Zacharias I (2012) Removal of phosphate from natural waters using innovative modified bentonites. Appl Clay Sci 62–63:101–106
Zamparas M, Drosos M, Georgiou Y, Deligiannakis Y, Zacharias I (2013) A novel bentonite-humic acid composite material Bephos for removal of phosphate and ammonium from eutrophic waters. Chem Eng J 225:43–51
Zhu G, Qin B (2003) Chemical sequential extraction of phosphorus in lake sediments. J Agro-Environ Sci 22:349–352 (In Chinese)
Acknowledgments
This research was supported by Tianjin Research Program of Application Foundation and Advanced Technology (Key project, No. 15JCZDJC40400), Tianjin, China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
(docx 15 kb)
Rights and permissions
About this article
Cite this article
Liu, S., Li, J., Yang, Y. et al. Influence of environmental factors on the phosphorus adsorption of lanthanum-modified bentonite in eutrophic water and sediment. Environ Sci Pollut Res 23, 2487–2494 (2016). https://doi.org/10.1007/s11356-015-5453-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-015-5453-z