Abstract
Background, aim and scope
Because of high discharged volumes and effluent composition, wastewater from the textile industry can be considered as the most polluting amongst all industrial sectors, thus greatly requiring appropriate treatment technologies. Although some abiotic methods for the reduction of several dyes exist, these require highly expensive catalysts and reagents. Biotechnological approaches were proven to be potentially effective in the treatment of this pollution source in an eco-efficient manner. The white-rot fungi are, so far, the most efficient microorganisms in degrading synthetic dyes. This white-rot fungi’s property is due to the production of extracellular lignin-modifying enzymes, which are able to degrade a wide range of xenobiotic compounds because of their low substrate specificity. In this paper, we studied the ability of the white-rot fungus Phanerochaete chrysosporium immobilised into Ca-alginate beads to decolourise different recalcitrant azo dyes such as Direct Violet 51 (DV), Reactive Black 5 (RB), Ponceau Xylidine (PX) and Bismark Brown R (BB) in successive batch cultures. To the best of our knowledge, this is the first study on the immobilisation of P. chrysosporium into Ca-alginate beads for its application in dye decolouration.
Materials and methods
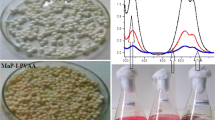
P. chrysosporium was immobilised into Ca-alginate beads using a method of gel recoating to minimise cellular leaking. The immobilised fungus was transferred to 250-ml Erlenmeyer flasks containing 50 ml of growth medium and incubated on an orbital shaker at 150 rpm and 30°C for 7 days. The ratio of beads/medium used was 10% (w/v). The dyes were added into the culture flasks when MnP production started (50 U l−1), which corresponded with the seventh cultivation day. MnP activity and dye decolouration were measured spectrophotometrically.
Results
The dyes DV, RB and PX were almost totally decolourised at the end of each batch during the course of three successive batches. However, the dye BB was more resistant to decolouration and it was not completely decolourised (86.7% in 144 h). Further, the beads were kept in sterilised calcium chloride (2 g l−1) for 3 weeks at 4°C. After these three storage weeks, the immobilised P. chrysosporium was again efficiently reused for azo dye decolouration during two successive batches, decolouration being more effective even for BB. Also, the in vitro decolouration of the aforementioned azo dyes by crude MnP from P. chrysosporium was performed. The decolouration levels obtained were lower than those attained with the whole cultures especially for RB and BB dyes, in spite of the fact that dye concentrations used were considerable lower.
Discussion
The good performance of the immobilisation system was likely due to the gel re-coating method utilised to prepare the alginate beads which not only maintained the beads integrity but also avoided cellular leaking. The lower decolouration percentages obtained by the enzyme indicates that the mycelial biomass may supply other intracellular or mycelial-bound enzymes, or other compounds that favour dye decolouration.
Conclusions
Immobilised P. chrysosporium efficiently decolourised different types of azo dyes. In this decolouration process, the MnP secreted by the fungus played the main role whilst adsorption was found to be negligible except for the dye BB.
Recommendations and perspectives
Efforts should be made to scale up and apply fungal decolouration techniques to real industrial dye-containing wastewater. Further, detailed characterisation of the intermediates and metabolites produced during biodegradation must be done to ensure the safety of the decolourised wastewater.






Similar content being viewed by others
References
Blánquez P, Casas N, Font X, Gabarrell X, Sarrà M, Caminal G, Vicent T (2004) Mechanism of textile metal dye biotransformation by Trametes versicolor. Water Res 38:2166–2172
Cohen Y (2001) Biofiltration—the treatment of fluids by microorganisms immobilized into the filter bedding material: a review. Bioresource Technol 77:257–274
Crini G (2006) Non-conventional low-cost adsorbents for dye removal: a review. Bioresource Technol 97:1061–1085
Cripps C, Bumpus JA, Aust SD (1990) Biodegradation of azo and heterocyclic dyes by Phanerochaete. Appl Environ Microbiol 56:1114–1118
Dosoretz CG, Chen HC, Grethlein ME (1990) Effect of oxygenation conditions on submerged cultures of Phanerochaete chrysosporium. Appl Microbiol Biotechnol 34:131–137
Ertugrul S, Bakır M, Donmez G (2008) Treatment of dye-rich wastewater by an immobilized thermophilic cyanobacterial strain: Phormidium sp. Ecol Eng 32:244–248
Glenn JK, Gold MH (1983) Decolorization of several polymeric dyes by the lignin-degrading basidiomycetes Phanerochaete chrysosporium. Appl Environ Microbiol 45:1741–1747
Hong H, Hwang S, Chang Y (2000) Biosorption of 1,2,3,4-tetrachlorodibenzo-p-dioxin and poly chlorinated dibenzofurans by Bacillus pulmilus. Water Resour 34:349–352
Kourkoutas Y, Bekatorou A, Banat IM, Marchant R, Koutinas AA (2004) Immobilization technologies and support materials suitable in alcohol beverages production: a review. Food Microbiol 21:377–397
Kuwahara M, Glenn JK, Morgar MA, Gold MH (1984) Separation and characterization of two extracellular H2O2 dependent oxidases from ligninolytic cultures of Phanerochaete chrysosporium. FEBS Lett 169:247–250
Laca A, Garcia LA, Diaz M (2000) Immobilization (locking) of Serratia marcescens in several supports. Afinidad 57:109–114
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Moldes D, Rodríguez Couto S, Cameselle C, Sanromán A (2003) Study of the degradation of dyes by MnP of Phanerochaete chrysosporium produced in a fixed-bed bioreactor. Chemosphere 51:295–303
Shin M, Nguyen T, Ramsay J (2002) Evaluation of support materials for the surface immobilization and decoloration of Amaranth by Trametes versicolor. Appl Microbiol Biotechnol 60:218–223
Sriamornsak P (1998) Preliminary investigation of some polysaccharides as a carrier for cell entrapment. Eur J Pharm Biopharm 46:233–236
Tien M, Kirk TK (1984) Lignin-degrading enzyme from Phanerochaete chrysosporium: purification, characterization and catalytic properties of a unique H2O2-requiring oxygenase. Proc Natl Acad Sci U S A 81:2280–2284
Tien M, Kirk TK (1988) Lignin peroxidase of Phanerochate chrysosporium. Methods Enzymol 16:238–249
Trupkin S, Levin L, Forchiassin F, Viale A (2003) Optimization of a culture medium for ligninolytic enzyme production and synthetic dye decolorization using response surface methodology. J Ind Microbiol Biotechnol 30:682–690
Yin L, Dan-Li X (2004) Decolorization and biodegradation of dye wastewaters by facultative-aerobic process. Env Sci Pollut Res 11:372–377
Acknowledgements
This research was supported by the Spanish Ministry of Education and Science (Project CTQ2007-66541). The authors would like to express their sincere thanks to Prof. Dr. D. Cullen for the kind gift of the P. chrysosporium strain and to Prof. Dr. José L. Toca-Herrera for kindly lending us the UV–Visible spectrophotometer (Thermo Spectronic, Cambridge, UK). SRC is a Ramón y Cajal Senior Research Fellow and gratefully acknowledges the Spanish Ministry of Education and Science for promoting the Ramón y Cajal Programme and the European Social Fund for co-financing her contract. KE thanks the University of Tehran (Iran) for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Enayatzamir, K., Alikhani, H.A., Yakhchali, B. et al. Decolouration of azo dyes by Phanerochaete chrysosporium immobilised into alginate beads. Environ Sci Pollut Res 17, 145–153 (2010). https://doi.org/10.1007/s11356-009-0109-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-009-0109-5