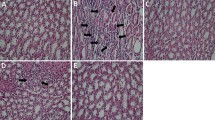
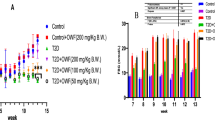
Abstract
Diabetes is a serious condition that is linked to the development of oxidative stress causing among many other effects, kidney failure and pancreatic disorders. However, traditional plant-based remedies can be considered an alternative to diabetes healing. In this context, this study was oriented towards evaluating the protective effect of the flowers of Opuntia microdasys Lehm. collected in Tunisia at a biochemical and histological level on kidneys and pancreas of a type 2 diabetic rats. Renal and pancreatic toxicities were induced in diabetic male Wistar rats by fructose alloxan. Diabetic rats were treated with an extract obtained from flowers collected at post-flowering stage (OFP) (100 and 200 mg kg−1 bw) and metformin (100 mg kg−1 bw) for 28 days. Oral administration of OFP at 200 mg kg−1 bw showed significant reduction of the uric acid, urea, creatinine, amylase, lipase, and glycated hemoglobin (HbAlc). The levels of SOD, CAT, and GPx were increased, while protein carbonyls and lipid peroxidation TBARS levels were reduced in the kidney and pancreas. The altered kidney and pancreas histology were restored in rats treated with OFP. Thus, the present study demonstrated that OFP has antihyperglycemic activity in fructose-alloxan-induced diabetic rats.



Similar content being viewed by others
References
Abd El-Razek FH, Hassan AA (2011) Nutritional value and hypoglycemic effect of prickly cactus pear (Opuntia ficus-indica) fruit juice in alloxan-induced diabetic rats. Aust J Basic Appl Sci 5:356–377
Abuyassin B, Laher I (2016) Diabetes epidemic sweeping the Arab world. World J Diabetes 7:165–174. https://doi.org/10.4239/wjd.v7.i8.165
Aebi H (1984) Catalase in vitro. In: Packer L (ed) Methods in enzymology. Academic Press, San Diego, pp 121–126
Alagammal M, Nishanthini A, Mohan VR (2012) Antihyperglycemic and antihyperlipidaemic effect of Polygala rosmarinifolia Wright & Arn on alloxan induced diabetic rats. J Appl Pharm Sci 2:143–148. https://doi.org/10.7324/JAPS.2012.2928
Alimi H, Hfaiedh N, Bouoni Z, Sakly M, Ben Rhouma K (2011) Evaluation of antioxidant and antiulcerogenic activities of Opuntia ficus indica f. inermis flowers extract in rats. Environ Toxicol Pharmacol 32:406–416. https://doi.org/10.1016/j.etap.2011.08.007
Ammar I, Ennouri M, Attia H (2015) Phenolic content and antioxidant activity of cactus (Opuntia ficus-indica L.) flowers are modified according to the extraction method. Ind Crop Prod 64:97–104. https://doi.org/10.1016/j.indcrop.2014.11.030
Balamurugan K, Nishanthini A, Mohan VR (2014) Antidiabetic and antihyperlipidaemic activity of ethanol extract of Melastoma malabathricum Linn. leaf in alloxan induced diabetic rats. Asian Pac J Trop Biomed 4:S442–S448. https://doi.org/10.12980/APJTB.4.2014C122
Behradmanesh S, Derees F, Kopaei MR (2013) Effect of Salvia officinalis on diabetic patients. J Ren Inj Prev 2:51–54. https://doi.org/10.12861/jrip.2013.18
Benayad Z, Martinez-Villaluenga C, Frias J, Gomez-Cordoves C, Es-Safi NE (2014) Phenolic composition, antioxidant and anti-inflammatory activities of extracts from Moroccan Opuntia ficus-indica flowers obtained by different extraction methods. Ind Crop Prod 62:412–420. https://doi.org/10.1016/j.indcrop.2014.08.046
Bouzidi A, Benzarti A, El AA et al (2016) Chemical composition , antioxidant and antimicrobial effects of Tunisian Limoniastrum guyonianum Durieu ex Boiss extracts. Pak J Pharm Sci 29:1299–1305
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Buege JA, Aust SD (1978) Microsomal lipid peroxidation. Methods Enzymol 52:302–10
Burski K, Ueland T, Maciejewski R (2004) Serum amylase activity disorders in the course of experimental diabetes in rabbits. Vet Med (Praha) 49:197–200
Chahdoura H, Barreira JCM, Barros L et al (2014) Phytochemical characterization and antioxidant activity of Opuntia microdasys (Lehm.) Pfeiff flowers in different stages of maturity. J Funct Foods 9:27–37. https://doi.org/10.1016/j.jff.2014.04.011
Chahdoura H, Adouni K, Khlifi A, Dridi I, Haouas Z, Neffati F, Flamini G, Mosbah H, Achour L (2017a) Hepatoprotective effect of Opuntia microdasys (Lehm.) Pfeiff flowers against diabetes type II induced in rats. Biomed Pharmacother 94:79–87. https://doi.org/10.1016/j.biopha.2017.07.093
Chahdoura H, El Bok S, Refifa T et al (2017b) Activity of anti-inflammatory, analgesic and antigenotoxic of the aqueous flower extracts of Opuntia microdasys Lem. Pfeiff. J Pharm Pharmacol 69:1056–1063. https://doi.org/10.1111/jphp.12734
Chandirasegaran G, Elanchezhiyan C, Ghosh K, Sethupathy S (2017) Berberine chloride ameliorates oxidative stress, inflammation and apoptosis in the pancreas of Streptozotocin induced diabetic rats. Biomed Pharmacother 95:175–185. https://doi.org/10.1016/j.biopha.2017.08.040
Crissman JW, Goodman DG, Hildebrandt PK, Maronpot RR, Prater DA, Riley JH, Seaman WJ, Thake DC (2004) Best practices guideline: toxicologic histopathology. Toxicol Pathol 32:126–131. https://doi.org/10.1080/01926230490268756
Dalle-Donne I, Rossi R, Giustarini D, Milzani A, Colombo R (2003) Protein carbonyl groups as biomarkers of oxidative stress. Clin Chim Acta 329:23–38. https://doi.org/10.1016/S0009-8981(03)00003-2
Ejaz AA, Mu W, Kang D-H et al (2007) Could uric acid have a role in acute renal failure? Clin J Am Soc Nephrol 2:16–21. https://doi.org/10.2215/CJN.00350106
El Arem A, Zekri M, Thouri A et al (2014) Oxidative damage and alterations in antioxidant enzyme activities in the kidneys of rat exposed to trichloroacetic acid: protective role of date palm fruit. J Physiol Biochem 70:297–309. https://doi.org/10.1007/s13105-013-0302-3
Fex M, Mulder H (2008) Lipases in the pancreatic beta-cell: implications for insulin secretion. Biochem Soc Trans 36:885–890. https://doi.org/10.1042/BST0360885
Flohé L, Gunzler AW (1984) Assays of glutathione peroxidase. Methods in enzymology, In, pp 114–120
Gao J, Han YL, Jin ZY, Xu XM, Zha XQ, Chen HQ, Yin YY (2015) Protective effect of polysaccharides from Opuntia dillenii Haw. fruits on streptozotocin-induced diabetic rats. Carbohydr Polym 124:25–34. https://doi.org/10.1016/j.carbpol.2015.01.068
Giacco F, Brownlee M (2010) Oxidative stress and diabetic complications. Circ Res 107:1058–1070. https://doi.org/10.1161/CIRCRESAHA.110.223545
Gupta RC, Chang D, Nammi S et al (2017) Interactions between antidiabetic drugs and herbs : an overview of mechanisms of action and clinical implications. Diabetol Metab Syndr 9:1–12. https://doi.org/10.1186/s13098-017-0254-9
Kang D-H, Nakagawa T, Feng L et al (2002) A role for uric acid in the progression of renal disease. J Am Soc Nephrol 13:2888–2897. https://doi.org/10.1097/01.ASN.0000034910.58454.FD
Kumar Kondeti V, Rao Badri K, Rajasekhar Maddirala D et al (2010) Effect of Pterocarpus santalinus bark, on blood glucose, serum lipids, plasma insulin and hepatic carbohydrate metabolic enzymes in streptozotocin-induced diabetic rats. Food Chem Toxicol 48:1281–1287. https://doi.org/10.1016/j.fct.2010.02.023
Laight DW, Carrier MJ, Änggård EE (2000) Antioxidants, diabetes and endothelial dysfunction. Cardiovasc Res 47:457–464. https://doi.org/10.1016/S0008-6363(00)00054-7
Levine RL (2002) Carbonyl modified proteins in cellular regulation, aging, and disease rodney. Free Radic Biol Med 32:797–803. https://doi.org/10.1016/S0891-5849(02)00780-3
Lysaght MJ (2002) Maintenance dialysis population dynamics: current trends and long-term implications. J Am Soc Nephrol 13:S37–S40
Mahjoubi-Samet A, Fetoui H, Zeghal N (2008) Nephrotoxicity induced by dimethoate in adult rats and their suckling pups. Pestic Biochem Physiol 91:96–103. https://doi.org/10.1016/j.pestbp.2008.01.009
Mercier Y, Gatellier P, Renerre M (2004) Lipid and protein oxidation in vitro, and antioxidant potential in meat from Charolais cows finished on pasture or mixed diet. Meat Sci 66:467–473. https://doi.org/10.1016/S0309-1740(03)00135-9
Missaoui S, Ben Rhouma K, Yacoubi M-T, Sakly M, Tebourbi O (2014) Vanadyl sulfate treatment stimulates proliferation and regeneration of beta cells in pancreatic islets. J Diabetes Res 2014:1–7. https://doi.org/10.1155/2014/540242
Naraharisetti SB, Aggarwal M, Ranganathan V, Sarkar SN, Kataria M, Malik JK (2009) Effects of simultaneous repeated exposure at high levels of arsenic and malathion on hepatic drug-biotransforming enzymes in broiler chickens. Environ Toxicol Pharmacol 28:213–218. https://doi.org/10.1016/j.etap.2009.04.006
Parving HH, Lewis JB, Ravid M, Remuzzi G, Hunsicker LG (2006) Prevalence and risk factors for microalbuminuria in a referred cohort of type II diabetic patients: a global perspective. Kidney Int 69:2057–2063. https://doi.org/10.1038/sj.ki.5000377
Rafieian-Kopaei M, Baradaran A (2013) Teucrium polium and kidney. J Ren Inj Prev 2:3–4. https://doi.org/10.12861/jrip.2013.02
Rains JL, Jain SK (2011) Oxidative stress, insulin signaling, and diabetes. Free Radic Biol Med 50:567–575. https://doi.org/10.1016/j.freeradbiomed.2010.12.006
Remuzzi G, Schieppati A, Ruggenenti P (2002) Nephropathy in patients with type 2 diabetes. N Engl J Med 346:1145–1151. https://doi.org/10.1056/NEJMcp011773
Ross SA, Gulve EA, Wang M (2004) Chemistry and biochemistry of type 2 diabetes. Chem Rev 104:1255–1282. https://doi.org/10.1021/CR0204653
Said S, Hernandez GT (2014) The link between chronic kidney disease and cardiovascular disease. J Nephropathol 3:99–104. https://doi.org/10.12860/jnp.2014.19
Sugden PH, Fuller SJ (1991) Regulation of protein turnover in skeletal and cardiac muscle. Biochem J 273:21–37. https://doi.org/10.1042/bj2730021
Sunmonu TO, Afolayan AJ (2013) Evaluation of antidiabetic activity and associated toxicity of Artemisia afra aqueous extract in Wistar rats. Evidence-based complement. Altern Med 2013:1–8. https://doi.org/10.1155/2013/929074
Viswanatha Swamy AHM, Wangikar U, Koti BC et al (2011) Cardioprotective effect of ascorbic acid on doxorubicin-induced myocardial toxicity in rats. Indian J Pharmacol 43:507–511. https://doi.org/10.4103/0253-7613.84952
Yang N, Zhao M, Zhu B, Yang B, Chen C, Cui C, Jiang Y (2008) Anti-diabetic effects of polysaccharides from Opuntia monacantha cladode in normal and streptozotocin-induced diabetic rats. Innov Food Sci Emerg Technol 9:570–574. https://doi.org/10.1016/j.ifset.2007.12.003
Yousef MI, Awad TI, Mohamed EH (2006) Deltamethrin-induced oxidative damage and biochemical alterations in rat and its attenuation by vitamin E. Toxicology 227:240–247. https://doi.org/10.1016/j.tox.2006.08.008
Zhao X, Pollock DM, Inscho EW, Zeldin DC, Imig JD (2003) Decreased renal cytochrome P450 2C enzymes and impaired vasodilation are associated with angiotensin salt-sensitive hypertension. Hypertension 41:709–714. https://doi.org/10.1161/01.HYP.0000047877.36743.FA
Ziani BEC, Calhelha RC, Barreira JCM, Barros L, Hazzit M, Ferreira ICFR (2015) Bioactive properties of medicinal plants from the Algerian flora: selecting the species with the highest potential in view of application purposes. Ind Crop Prod 77:582–589. https://doi.org/10.1016/j.indcrop.2015.09.034
Ziani BEC, Barros L, Boumehira AZ, Bachari K, Heleno SA, Alves MJ, Ferreira ICFR (2018) Profiling polyphenol composition by HPLC-DAD-ESI/MSn and the antibacterial activity of infusion preparations obtained from four medicinal plants. Food Funct 9:149–159. https://doi.org/10.1039/C7FO01315A
Acknowledgments
We would like to thank Dr. Habib Mosbah for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declares that they have no conflict of interest.
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Chahdoura, H., Khlifi, A., Lamine, J.B. et al. Protective potential of Opuntia microdasys flower decoction on fructose-alloxan-induced diabetic rats on kidney and pancreas: chemical and immunohistochemical analyses. Environ Sci Pollut Res 25, 33645–33655 (2018). https://doi.org/10.1007/s11356-018-3290-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-3290-6