Summary
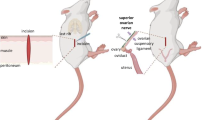
The role of ovarian adrenergic nerves in follicular growth was studied in prepubertal guinea pigs by determining the effect of sympathectomy on 1) follicle populations and 2) follicular development following exogenous gonadotropin administration. Selective unilateral ovarian sympathectomy was achieved by injecting 6-hydroxydopamine into a surgically closed periovarian bursa on day 20 postpartum. The contralateral surgically closed ovarian bursa was injected with the vehicle used for 6-hydroxydopamine. On day 25, animals were injected with pregnant mare serum or saline followed by human chorionic gonadotropin or saline 48 h later. All animals were laparotomized on day 28 and blood from utero-ovarian veins was collected bilaterally for androstenedione determination. Ovaries were processed for morphometric analysis of follicles. The sympathectomized ovary in saline-injected animals had a significant decrease in preantral follicles (characterized by ≤2 layers of granulosa cells without antrum formation), an increase in 310–500 μm diameter atretic follicles and an increase in follicles ≥700 um compared to the contralateral control ovary. There were no differences in androstenedione levels from the two sides, ovarian weights or the total number of follicles per ovary. Neither ovary had corpora lutea. The sympathectomized ovary in animals injected with gonadotropins was not different from the contralateral ovary in any of the parameters measured. Both control and sympathectomized ovaries had newly formed corpora lutea in response to the exogenous gonadotropins. These results suggest that ovarian adrenergic nerves normally participate in follicular development in the prepubertal guinea pig. However, exogenous gonadotropins may override neural influences on the prepubertal ovary.
Similar content being viewed by others
References
Azzolin GC, Saiduddin S (1983) Effects of androgens on the ovarian morphology of the hypophysectomized rat. Proc Soc Exp Med 172:70–73
Burden HW (1972) Adrenergic innervation in the ovaries of the rat and guinea pig. Am J Anat 133:455–462
Curry TE Jr, Lawrence IE Jr, Burden HW (1984) Ovarian sympathectomy in the guinea pig: I. Effects on follicular development during the estrous cycle. Cell Tissue Res 236:257–263
Dohm GL, Louis TM (1978) Changes in androstenedione, testosterone and protein metabolism as a result of exercise. Proc Soc Exp Biol Med 158:622–625
Donovan BT, ter Haar MB, Lockhart AN, Mackinnon PCB, Mattock JM, Peddie MJ (1975a) Changes in the concentration of luteinizing hormone in plasma during development in the guinea pig. J Endocrinol 64:511–520
Donovan BT, ter Haar MB, Lockhart AN, Peddie MJ (1975b) Changes in the concentration of follicle-stimulating hormone in plasma during development in the guinea pig. J Endocrinol 64:521–528
Fink G, Schofield GC (1971) Experimental studies on the innervation of the ovary in cats. J Anat 109:115–126
Hamburger C, Pedersen-Bjergaard K (1946) On the effect of gonadotropins in normal and infantile female guinea pigs. Acta Pathol Microbiol Scand 23:85–101
Lawrence IE Jr, Louis TM, Burden HW (1982) The effects of 6-hydroxydopamine administration during the prepubertal period on the guinea pig ovary. Anat Rec 202:109A
Louvet JP, Harman SM, Schreiber JR, Ross GT (1975) Evidence for a role of androgens in follicular maturation. Endocrinology 97:366–372
Mills PG, Reed M (1971) The onset of first oestrus in the guinea pig and the effects of gonadotropins and oestradiol in the immature animal. J Endocrinol 50:329–337
Peddie MJ (1978) Follicular development in the immature female guinea pig: Effects of human chorionic gonadotropin on morphological development and steroid secretion in vivo and in vitro. Eur J Obstet Gynecol Reprod Biol 9:204
Peddie MJ (1980) Follicular development in the immature guinea pig. J Endocrinol 84:323–331
Peluso JJ, Steger RW, Hafez ESE (1977) Sequential changes associated with the degeneration of preovulatory rat follicles. J Reprod Fert 49:215–218
Perry JS, Rowlands IW (1963) Hypophysectomy of the immature guinea pig and the ovarian response to gonadotropins. J Reprod Fertilo 6: 393–404
Rawson JMR, Galey CE, Weinbrug LC, Hodgson BJ (1979) Effects of gonadotropins on follicular development, ovulation, and atresia in the mature guinea pig. Hormone Res 10:25–36
Richards JS (1980) Maturation of ovarian follicles: actions and interactions of pituitary and ovarian hormones on follicular cell differentiation. Physiol Rev 60:51–88
Tranzer JP, Thoenen H (1967) Ultra-morphologische Veränderungen der sympathischen Nervendigungen der Katze nach Vorbehandlung mit 5- und 6-Hydroxydopamine. Naunyn-Schmiedebergs Arch Pharmakol Exp Pathol 257:343–344
Zar JH (1974) Biostatistical analysis. Prentice Hall, Inc., New Jersey
Zarrow MX, Quinn DL (1963) Superovulation in the immature rat following treatment with PMS alone and inhibition of PMSinduced ovulation. J Endocrinol 26:181–188
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Curry, T.E., Lawrence, I.E. & Burden, H.W. Ovarian sympathectomy in the guinea pig. Cell Tissue Res. 236, 593–596 (1984). https://doi.org/10.1007/BF00217227
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00217227