Abstract
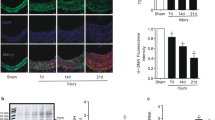
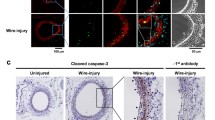
Nitric oxide (NO) limits formation of neointimal hyperplasia in animal models of arterial injury in large part by inhibiting vascular smooth muscle cell (VSMC) proliferation through cell cycle arrest. The ubiquitin-conjugating enzyme UbcH10 is responsible for ubiquitinating cell cycle proteins for proper exit from mitosis. We hypothesize that NO prevents VSMC proliferation, and hence neointimal hyperplasia, by decreasing levels of UbcH10. Western blotting and immunofluorescent staining showed that NO reduced UbcH10 levels in a concentration-dependent manner in VSMC harvested from the abdominal aortas of Sprague-Dawley rats. Treatment with NO or siRNA to UbcH10 decreased both UbcH10 levels and VSMC proliferation (P < 0.001), while increasing UbcH10 levels by plasmid transfection or angiotensin II stimulation increased VSMC proliferation to 150% (P = 0.008) and 212% (P = 0.002) of control, respectively. Immunofluorescent staining of balloon-injured rat carotid arteries showed a ~4-fold increase in UbcH10 levels, which was profoundly decreased following treatment with NO. Western blotting of carotid artery lysates showed no UbcH10 in uninjured vessels, a substantial increase in the injury alone group, and a significant decrease in the injury + NO group (~3-fold reduction versus injury alone). Importantly, in vitro and in vivo, a marked increase in polyubiquitinated UbcH10 was observed in the NO-treated VSMC and carotid arteries, respectively, indicating that NO may be decreasing unmodified UbcH10 levels by increasing its ubiquitination. Central to our hypothesis, we report that NO decreases UbcH10 levels in VSMC in vitro and following arterial injury in vivo in association with increasing polyubiquitinated-UbcH10 levels. These changes in UbcH10 levels correlate with VSMC proliferation and neointimal hyperplasia, making UbcH10 a promising therapeutic target for inhibiting this proliferative disease.







Similar content being viewed by others
References
Rape, M., & Kirschner, M. W. (2004). Autonomous regulation of the anaphase-promoting complex couples mitosis to S-phase entry. Nature, 432(7017), 588–595.
Rape, M., Reddy, S. K., & Kirschner, M. W. (2006). The processivity of multiubiquitination by the APC determines the order of substrate degradation. Cell, 124(1), 89–103.
Hershko, A., & Ciechanover, A. (1998). The ubiquitin system. Annual Review of Biochemistry, 67, 425–479.
Crews, C. M. (2003). Feeding the machine: Mechanisms of proteasome-catalyzed degradation of ubiquitinated proteins. Current Opinion in Chemical Biology, 7(5), 534–539.
De, G. A., Ganier, O., & Cohen-Fix, O. (2006). Before and after the spindle assembly checkpoint—an APC/C point of view. Cell Cycle, 5(18), 2168–2171.
Berlingieri, M. T., Pallante, P., Sboner, A., Barbareschi, M., Bianco, M., Ferraro, A., et al. (2007). UbcH10 is overexpressed in malignant breast carcinomas. European Journal of Cancer, 43(18), 2729–2735.
Okamoto, Y., Ozaki, T., Miyazaki, K., Aoyama, M., Miyazaki, M., & Nakagawara, A. (2003). UbcH10 is the cancer-related E2 ubiquitin-conjugating enzyme. Cancer Research, 63(14), 4167–4173.
Wagner, K. W., Sapinoso, L. M., El-Rifai, W., Frierson, H. F., Butz, N., Mestan, J., et al. (2004). Overexpression, genomic amplification and therapeutic potential of inhibiting the UbcH10 ubiquitin conjugase in human carcinomas of diverse anatomic origin. Oncogene, 23(39), 6621–6629.
Shears, L. L., Kibbe, M. R., Murdock, A. D., Billiar, T. R., Lizonova, A., Kovesdi, I., et al. (1998). Efficient inhibition of intimal hyperplasia by adenovirus-mediated inducible nitric oxide synthase gene transfer to rats and pigs in vivo. Journal of the American College of Surgeons, 187(3), 295–306.
Pearce, C. G., Najjar, S. F., Kapadia, M. R., Murar, J., Eng, J., Lyle, B., et al. (2008). Beneficial effect of a short-acting NO donor for the prevention of neointimal hyperplasia. Free Radical Biology and Medicine, 44(1), 73–81.
Kapadia, M. R., Chow, L. W., Tsihlis, N. D., Ahanchi, S. S., Eng, J. W., Murar, J., et al. (2008). Nitric oxide and nanotechnology: A novel approach to inhibit neointimal hyperplasia. Journal of Vascular Surgery, 47(1), 173–182.
Yu, S. M., Hung, L. M., & Lin, C. C. (1997). cGMP-elevating agents suppress proliferation of vascular smooth muscle cells by inhibiting the activation of epidermal growth factor signaling pathway. Circulation, 95(5), 1269–1277.
Kibbe, M. R., Li, J., Nie, S., Watkins, S. C., Lizonova, A., Kovesdi, I., et al. (2000). Inducible nitric oxide synthase (iNOS) expression upregulates p21 and inhibits vascular smooth muscle cell proliferation through p42/44 mitogen-activated protein kinase activation and independent of p53 and cyclic guanosine monophosphate. Journal of Vascular Surgery, 31(6), 1214–1228.
Chamley, J. H., Campbell, G. R., McConnell, J. D., & Groschel-Stewart, U. (1977). Comparison of vascular smooth muscle cells from adult human, monkey and rabbit in primary culture and in subculture. Cell and Tissue Research, 177(4), 503–522.
Hrabie, J. A., Klose, J. R., Wink, D. A., & Keefer, L. K. (1993). New nitric oxide-releasing zwitterions derived from polyamines. Journal of Organic Chemistry, 58(6), 1472–1476.
Kibbe, M. R., Nie, S., Seol, D. W., Kovesdi, I., Lizonova, A., Makaroun, M., et al. (2000). Nitric oxide prevents p21 degradation with the ubiquitin-proteasome pathway in vascular smooth muscle cells. Journal of Vascular Surgery, 31(2), 364–374.
Ahanchi, S. S., Varu, V. N., Tsihlis, N. D., Martinez, J., Pearce, C. G., Kapadia, M. R., et al. (2008). Heightened efficacy of nitric oxide-based therapies in type II diabetes mellitus and metabolic syndrome. American Journal of Physiology - Heart and Circulatory Physiology, 295(6), H2388–H2398.
Butz, N., Ruetz, S., Natt, F., Hall, J., Weiler, J., Mestan, J., et al. (2005). The human ubiquitin-conjugating enzyme Cdc34 controls cellular proliferation through regulation of p27Kip1 protein levels. Experimental Cell Research, 303(2), 482–493.
Banerjee, A., Gregori, L., Xu, Y., & Chau, V. (1993). The bacterially expressed yeast CDC34 gene product can undergo autoubiquitination to form a multiubiquitin chain-linked protein. Journal of Biological Chemistry, 268(8), 5668–5675.
Osaka, F., Kawasaki, H., Aida, N., Saeki, M., Chiba, T., Kawashima, S., et al. (1998). A new NEDD8-ligating system for cullin-4A. Genes and Development, 12(15), 2263–2268.
Ahanchi, S. S., Tsihlis, N. D., & Kibbe, M. R. (2007). The role of nitric oxide in the pathophysiology of intimal hyperplasia. Journal of Vascular Surgery, 45(Suppl A), A64–A73.
Meiners, S., Laule, M., Rother, W., Guenther, C., Prauka, I., Muschick, P., et al. (2002). Ubiquitin-proteasome pathway as a new target for the prevention of restenosis. Circulation, 105(4), 483–489.
Kapadia, M. R., Eng, J. W., Jiang, Q., Stoyanovsky, D. A., & Kibbe, M. R. (2009). Nitric oxide regulates the 26S proteasome in vascular smooth muscle cells. Nitric Oxide, 20(4), 279–288.
Faries, P. L., Rohan, D. I., Wyers, M. C., Marin, M. L., Hollier, L. H., Quist, W. C., et al. (2001). Relationship of the 20S proteasome and the proteasome activator PA28 to atherosclerosis and intimal hyperplasia in the human vascular system. Annals of Vascular Surgery, 15(6), 628–633.
Herrmann, J., Saguner, A. M., Versari, D., Peterson, T. E., Chade, A., Olson, M., et al. (2007). Chronic proteasome inhibition contributes to coronary atherosclerosis. Circulation Research, 101(9), 865–874.
Weekes, J., Morrison, K., Mullen, A., Wait, R., Barton, P., & Dunn, M. J. (2003). Hyperubiquitination of proteins in dilated cardiomyopathy. Proteomics, 3(2), 208–216.
Predmore, J. M., Wang, P., Davis, F., Bartolone, S., Westfall, M. V., Dyke, D. B., et al. (2010). Ubiquitin proteasome dysfunction in human hypertrophic and dilated cardiomyopathies. Circulation, 121(8), 997–1004.
Versari, D., Herrmann, J., Gossl, M., Mannheim, D., Sattler, K., Meyer, F. B., et al. (2006). Dysregulation of the ubiquitin-proteasome system in human carotid atherosclerosis. Arteriosclerosis, Thrombosis, and Vascular Biology, 26(9), 2132–2139.
Stone, D. H., Sivamurthy, N., Contreras, M. A., Fitzgerald, L., LoGerfo, F. W., & Quist, W. C. (2001). Altered ubiquitin/proteasome expression in anastomotic intimal hyperplasia. Journal of Vascular Surgery, 34(6), 1016–1022.
Acknowledgments
The authors would like to express their thanks to the Northwestern University Institute for BioNanotechnology in Medicine, the Northwestern University Feinberg Cardiovascular Research Institute, to Lynnette Dangerfield for her administrative support, and to Edwards Lifesciences for providing the Fogarty balloon catheters. This work was supported in part by funding from the National Institutes of Health (1K08HL0842-03 to MRK), the American Vascular Association (Mentored Clinical Scientist Development Award to MRK), the American Heart Association (0725766Z and 09POST2230028 to NDT), and by the generosity of Mrs. Hilda Rosenbloom and Mrs. Eleanor Baldwin. In addition, part of this research was supported with federal funds from the National Cancer Institute, NIH, under Contract N01-CO-2008-00001 with SAIC-Frederick, Inc. and by the Intramural Research Program of the NIH, National Cancer Institute, Center for Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsihlis, N.D., Oustwani, C.S., Vavra, A.K. et al. Nitric Oxide Inhibits Vascular Smooth Muscle Cell Proliferation and Neointimal Hyperplasia by Increasing the Ubiquitination and Degradation of UbcH10. Cell Biochem Biophys 60, 89–97 (2011). https://doi.org/10.1007/s12013-011-9179-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-011-9179-3