Abstract
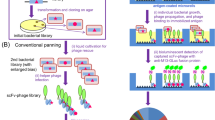
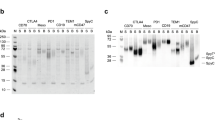
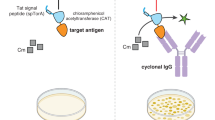
We have applied the combinatorial immunoglobulin library and phage display technologies to generate monoclonal rabbit single-chain Fv (scFv) antibody fragments specific for recombinant human leukemia inhibitory factor (rhLIF). The B cell immunoglobulin repertoire of an immunized rabbit was immortalized by the combinatorial cloning of the rearranged variable domains of light (VL) and heavy (VH) chains. Affinity selection of the library displaying the rabbit antibody domains on the phage surface resulted in the isolation of phage encoding scFv antibodies which specifically bind to the antigen. We utilized the methylotrophic yeast Pichia pastoris for high level secretion of soluble and functional scFv antibody fragment. More than 100 mg/L of pure and functional rabbit anti-rhLIF scFv antibody was obtained directly from the P. pastoris culture supernatant by one-step affinity chromatography.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Barbas, C.F. III. 1993. Recent advances in phage display. Curr. Opin. Biotechnol. 4: 526–530.
Winter, G., Griffiths, A.D., Hawkins, R.E., Hoogenboom, H.R. 1994. Making antibodies by phage display technology. Annu. Rev. Immunol. 12: 433–455.
Huse, W.D., Sastry, L., Iverson, S.A., Kang, A.S., Alting-Mees, M., Burton, D.R., Benkovic, S.J. and Lerner, R.A. 1989. Generation of a large combinatorial library of the immunoglobulin repertoire in phage lambda . Science 246: 1275–1281.
Clackson, T., Hoogenboom, H.R., Griffiths, A.D., Winter, G. 1991. Making antibody fragments using phage display libraries. Nature 352: 624–628.
Burton, D.R., Barbas, C.F. III, Persson, M.A., Koenig, S., Chanock, R.M., Lerner, R.A. 1991. A large array of human monoclonal antibodies to type 1 human immunodeficiency virus. from combinatorial libraries of asymptomatic seropositive individuals. Proc. Natl. Acad. Sci. USA 88: 10134–10137.
Barbas, C.F. III., Crowe, J.E., Cababa, D., Jones, T.M., Zebedee, S.L., Murphy, B.R., Chanock, R.M., Burton, D.R. 1992. Human monoclonal Fab fragments derived from a combinatorial library bind to respiratory syncytial virus F glycoprotein and neutralise infectivity. Proc. Natl. Acad. Sci. USA 89: 10164–10168.
Zebedee, S.L., Barbas, C.F. III., Hom, Y., Caothien R.H., Graff, R., Degraw, J., Pyati, J., LaPolla, R., Burton, D.R., Lerner, R.A., Thornton, G.B. 1992. Human combinatorial antibody libraries to hepatitis B surface antigen. Proc. Natl. Acad. Sci. USA 89: 3175–3179.
Williamson, R.A., Burioni, R., Sanna, P.P., Partridge, L.J., Barbas, C.F. III Burton, D.R . 1993. Human monoclonal antibodies against a plethora of viral pathogens from single combinatorial libraries. Proc. Natl. Acad. Sci. USA 90: 4141–4145.
Burioni, R., Williamson, R.A., Sanna, P.P., Bloom, F.E. and Burton, D.R. 1994. Recombinant human Fab to glycoprotein D neutralizes infectivity and prevents cell-to-cell transmission of herpes simplex viruses 1 and 2 in vitro . Proc. Natl. Acad. Sci. USA 91: 355–359.
Marks, J.D., Hoogenboom, H.R., Bonnert, T.P., McCafferty, J., Griffiths, A.D., Winter, G. 1991. By-passing immunization. Human antibodies from V-gene libraries displayed on phage. J. Mol. Biol. 222: 581–597.
Griffiths, A.D., Malmqvist, M., Marks, J.D., Bye, J.M., Embleton, M.J., McCafferty, J., Baier, M., Holliger, K.P., Gorick, B.D., Hughes-Jones, N.C., Hoogenboom, H.R. and Winter, G. 1993. Human anti-self antibodies with high specificity from phage display libraries. EMBO J. 12: 725–734.
Raman, C., Spieker-Polet, H., Yam, P.C., Knight, K.L. 1994. Preferential VH gene usage in rabbit Ig-secreting heterohybridomas. J. Immunol. 152: 3935–3945.
Patterson, P.H. 1994. Leukemia inhibitory factor, a cytokine at the interface between neurobiology and immunology. Proc. Natl. Acad. Sci. USA 91: 7833–7835.
Plückthun, A. 1991. Antibody engineering: advances from the use of Escherichia coli expression systems. Bio/Technology 9: 545–551.
Pack, P., Kujau, M., Schroeckh, V., Knüpfer, U., Wenderoth, R., Riesenberg, D., Plückthun, A. 1993. Improved bivalent miniantibodies, with identical avidity as whole antibodies, produced by high cell density fermentation of Escherichia coli . Biol. Technology 11: 1271–1277.
Carter, P., Kelley, R.F., Rodrigues, M.L., Snedecor, B., Covarrubias, M., Velligan, M.D., Wong, W.L.T., Rowland, A.M., Kotts, C.E., Carver, M.E., Yang, M., Bourell, J.H., Shephard, H.M. and Henner, D. 1992. High level Escherichia coli expression and production of a bivalent humanized antibody fragment. Biol Technology 10: 163–167.
Skerra, A. 1993. Bacterial expression of immunoglobulin fragments. Curr. Opin. Immunol. 5: 256–262.
Cregg, J.M., Madden, K.R. 1988. Development of the methylotrophic yeast, Pichia pastoris, as a host system for the production of foreign proteins. Dev. Ind. Microbiol. 29: 33–41.
Clare, J.J., Rayment, F.B., Ballantine, S.P., Sreekrishna, K., Romanos, M.A. 1991. High-level expression of tetanus toxin fragment C in Pichia pastoris strains containing multiple tandem integrations of the gene. Bio/Technology 9: 455–460.
Tschopp, J.F., Sverlow, G., Kosson, R., Craig, W. and Grinna, L. 1987. High-level secretion of glycosylated invertase in the methylotrophic yeast,Pichia pastoris. Bio/Technology 5: 1305–1308.
Digan, M.E., Lair, S.V., Brierley, R.A., Siegel, R.S., Williams, M.E., Ellis, S.B., Kellaris, P.A., Provow, S.A., Craig, W.S., Velicelebi, G., Harpold, M.M. and Thill, G.P. 1989. Continuous production of a novel lysozyme via secretion from the yeast, Pichia pastoris . Bio/Technology 7: 160–164.
Cregg, J.M., Vedvick, T.S., Raschke, W.C. 1993. Recent advances in the expression of foreign gene in Pichia pastoris . Bio/Technology 11: 905–910.
Kabat, E.A., Wu, T.T., Perry, H.M., Gottesman, K.S. and Foeller, C. 1991. Sequences of proteins of immunological interest. 5th edition, NIH publication 91–3242.
Barbas, C.F. III., Kang, A.S., Lerner, R.A . Benkovic, S.J. 1991. Assembly of combinatorial antibody libraries on phage surfaces: the geneIII site. Proc. Natl. Acad. Sci. USA 88: 7978–7982.
McCafferty, J., Griffiths, A.D., Winter, G. and Chiswell, D.J. 1990. Phage antibodies: filamentous phage displaying antibody variable domains. Nature 348: 552–554.
Gram, H., Strittmatter, U., Lorenz, M., Glück D. and Zenke, G. 1993. Phage display as a rapid gene expression system: production of bioactive cytokine-phage and generation of neutralizing monoclonal antibodies. J. Immunol. Meth. 161: 169–176.
Malmqvist, M. . 1992. Surface plasmon resonance for detection and measurement of antibody-antigen affinity and kinetics. Curr. Opin. Immunol. 5: 282–286.
Clare, J.J., Romanos, M.A., Rayment, F.B., Rowedder, J.E., Smith, M.A., Payne, M.M., Sreekrishna, K. and Henwood, C.A . 1991. Production of mouse epidermal growth factor in yeast: high-level secretion using Pichia pastoris strains containing multiple gene copies. Gene 105: 205–212
Lokker, N.A., Strittmatter, U., Steiner, C., Fagg, B., Graff, P., Kocher, H.P. and Zenke, G. 1991. Mapping the epitopes of neutralizing anti-human IL-3 monoclonal antibodies. Implications for structure-activity relationship. J. Immunol. 146: 893–898.
Skerra, A., Pfitzinger, I. and Plühun, A. . 1991. The functional expression of antibody Fv fragments in Escherichia coli: improved vectors and a generally applicable purification technique. Bio/Technology 9: 273–278.
Suter, M., Blaser, K., Aeby, P. and Crameri, R. 1992. Rabbit single domain antibodies specific to protein C expressed in prokaryotes. Immunol. Lett. 33: 53–60.
Knight, K.L. 1992 Restricted VH gene usage and generation of antibody diversity in rabbit. Annu. Rev. Immunol. 10: 593–616.
Becker, R.S. and Knight, K.L. 1990. Somatic diversification of immunoglobulin heavy chain VDJ genes: evidence for somatic gene conversion in rabbits. Cell 63: 987–997.
Knight, K.L. and Becker, R.S . 1990. Molecular basis of the allelic inheritance of rabbit immunoglobulin VH allotypes: implications for the generation of antibody diversity. Cell 60: 963–970.
Heidmann, O. and Rougeon, F . 1983. Diversity in the rabbit immunoglobulin kappa chain variable regions is amplified by nucleotide deletions and insertions at the V-J junction. Cell 34: 767–777.
Emorine, L., Dreher, K., Kindt, T.J. and Max, E.E. . 1983. Rabbit immunoglobulin k genes: structure of a germline b4 allotype J–C locus and evidence for several b4–related sequences in the rabbit genome. Proc. Natl. Acad. Sci. USA 80: 5709–5713.
Akimenko, M.A., Mariame, B., Rougeon, F . 1986. Evolution of the immunoglobulin k light chain locus in the rabbit: evidence for differential gene conversion events. Proc. Natl. Acad. Sci. USA 83: 5180–5183.
Hawkins, R.E., Russell, S.J., Winter, G . 1992. Selection of phage antibodies by binding affinity mimicking affinity maturation. J. Mol. Biol. 226: 889–896.
Studier, F.W., Rosenberg, A.H., Dunn, J. and Dubendorff, J.W. . 1990 Use of 77RNA polymerase to direct expression of cloned genes. Methods in Enzymol. 185: 60–89.
Evan, G.I., Lewis, G.K., Ramsay, G., Bishop, J.M. 1985. Isolation of monoclonal antibodies specific for human c–myc proto–oncogene product. Mol. Cell. Biol. 5: 3610–3616.
Ausubel, F.M., Brent, R., Kingston, R.E., Moore, D.D., Seidman, J.G., Smith, J.A., Struhl, K. 1991. Current Protocols in Molecular Biology. John Wiley & Sons, New York.
Chomczynski, P. and Sacch, N. 1987. Single–step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction. Anal. Biochem. 162: 156–159.
Jonsson, U., Fagerstam, L., Ivarson, B., Johnsson, B., Karlsson, R., Lundh, K., Lofas, S., Persson, B., Roos, H., Ronnberg, I., Sjolander, S., Stenberg, E., Stahlberg, R., Urbaniczky, C., Ostlin, H. . Malmquist, M. 1991. Real–time biospecific interaction analysis using surface plasmon resonance and a sensor chip technology. Biotechniques 11: 520–527.
Karlsson, R., Michaelsson, A. and Mattsson, L. . 1991. Kinetic analysis of monoclonal antibody-antigen interactions with a new biosensor based analytical system. J. Immunol. Methods 145: 229–240.
Cregg, J.M., Barringer, K.J., Hessler, A.Y. and Madden, K.R. 1985 Pichia pastoris as a hostsystem for transformations. Mol. Cell. Biol. 5: 3376–3385.
Colcher, D., Bird, R., Roselli, M., Hardman, K.D., Johnson, S., Pope, S., Dodd, S.W., Pantoliano, M.W., Milenic, D.E. . Schlom, J. 1990. In vivo tumor targeting of a recombinant single–chain antigen–binding protein. J. Natl. Cancer Inst. 82: 1191–1197.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ridder, R., Schmitz, R., Legay, F. et al. Generation of Rabbit Monoclonal Antibody Fragments from a Combinatorial Phage Display Library and Their Production in the Yeast Pichia pastoris. Nat Biotechnol 13, 255–260 (1995). https://doi.org/10.1038/nbt0395-255
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0395-255
This article is cited by
-
Efficient production of single-chain fragment variable-based N-terminal trimerbodies in Pichia pastoris
Microbial Cell Factories (2014)
-
High efficient expression of a functional humanized single-chain variable fragment (scFv) antibody against CD22 in Pichia pastoris
Applied Microbiology and Biotechnology (2014)
-
Covalently dimerized Camelidae antihuman TNFa single-domain antibodies expressed in yeast Pichia pastoris show superior neutralizing activity
Applied Microbiology and Biotechnology (2013)
-
Rapid production of antigen-specific monoclonal antibodies from a variety of animals
BMC Biology (2012)
-
Antibody production with yeasts and filamentous fungi: on the road to large scale?
Biotechnology Letters (2007)