Abstract
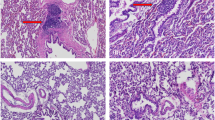
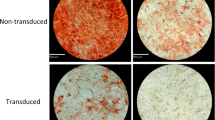
The objective of the study was to analyze and compare the abilities of various human cell types with inherently dissimilar osteogenic potentials to induce heterotopic bone formation following ex vivo transduction with two distinct adenoviral vectors encoding bone morphogenetic protein type 2 (BMP2). The cells comprised primary human bone marrow mesenchymal stem cells (BM-MSCs), primary human skin fibroblasts (SFs), and a human diploid fetal lung cell line (MRC-5). The vectors included adenovirus type 5 or a chimeric adenovirus type 5 with the fiber gene of adenovirus type 35 (Ad5F35-BMP2), both demonstrating significantly different expression of BMP2 in vitro. The experimental groups consisted of the three human cell types transduced with each of the two adenoviral vectors. Using nonobese diabetic severe combined immunodeficiency (NOD/SCID) mice, the transduced cells were injected intramuscularly following ex vivo adenoviral transduction. The nature and extent of heterotopic bone formation were analyzed radiographically and histologically. At 14 days postinjection, abundant, highly mineralized bone was formed in mice injected with Ad5F35-BMP2-transduced cells irrespective of the cell type. There was no statistically significant difference in the amount of bone formed between BM-MSCs, SFs, and MRC-5 cells transduced with Ad5F35-BMP2, as assessed from bone surface area on biplanar plain radiography. Substantially lesser amounts or no bone could be detected in mice injected with cells transduced with Ad5-BMP2. Immunohistochemical analysis confirmed the presence of human cells in muscle as early as 2 days postdelivery; however, at 6–7 days after injection, the transduced cells could not be detected in surrounding muscle, or in the heterotopic bone, indicating the host origin of the newly formed bone. The results of the study demonstrate no significant difference in osteoinductive properties between BM-MSCs, SFs, and MRC-5 cells transduced ex vivo with the same type of adenovirus encoding BMP2. The level of BMP2 expression appears to be a crucial factor determining the extent of heterotopic bone formation and was significantly affected by the type of adenovirus used. In the cell types studied, Ad5F35-BMP2 was more efficacious than Ad5-BMP2 in providing adequate levels of BMP2 for efficient osteoinduction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Lieberman JR et al. Regional gene therapy with a BMP-2-producing murine stromal cell line induces heterotopic and orthotopic bone formation in rodents. J Orthop Res 1998; 16: 330–339.
Riew KD et al. Induction of bone formation using a recombinant adenoviral vector carrying the human BMP-2 gene in a rabbit spinal fusion model. Calcif Tissue Int 1998; 63: 357–360.
Alden TD et al. In vivo endochondral bone formation using a bone morphogenetic protein 2 adenoviral vector. Hum Gene Ther 1999; 10: 2245–2253.
Krebsbach PH, Gu K, Franceschi RT, Rutherford RB . Gene therapy-directed osteogenesis: BMP-7-transduced human fibroblasts form bone in vivo. Hum Gene Ther 2000; 11: 1201–1210.
Varady P et al. Morphologic analysis of BMP-9 gene therapy-induced osteogenesis. Hum Gene Ther 2001; 12: 697–710.
Okubo Y et al. In vitro and in vivo studies of a bone morphogenetic protein-2 expressing adenoviral vector. J Bone Joint Surg 2001; 83A (Suppl 1): S99–S104.
Engstrand T et al. Transient production of bone morphogenetic protein 2 by allogeneic transplanted transduced cells induces bone formation. Human Gene Ther 2000; 11: 205–211.
Lee JY et al. Enhancement of bone healing based on ex vivo gene therapy using human muscle-derived cells expressing bone morphogenetic protein 2. Hum Gene Ther 2002; 13: 1201–1211.
Gazit D et al. Engineered pluripotent mesenchymal cells integrate and differentiate in regenerating bone: a novel cell-mediated gene therapy. J Gene Med 1999; 1: 121–133.
Musgrave DS et al. Ex vivo gene therapy to produce bone using different cell types. Clin Orthop 2000; 378: 290–305.
Olmsted EA et al. Adenovirus-mediated BMP2 expression in human bone marrow stromal cells. J Cell Biochem 2001; 82: 11–21.
Olmsted-Davis EA et al. Use of a chimeric adenovirus vector enhances BMP2 production and bone formation. Hum Gene Ther 2002; 13: 1337–1347.
Jaiswal N, Haynesworth SE, Caplan AI, Bruder SP . Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. J Cell Biochem 1997; 64: 295–312.
Davis AR et al. Construction of adenoviral vectors. Mol Biotechnol 2001; 18: 63–70.
Gutekunst CA et al. The cellular and subcellular localization of huntingtin-associated protein 1 (HAP1): comparison with huntingtin in rat and human. J Neurosci 1998; 18: 7674–7686.
Farinas I, Yoshida CK, Backus C, Reichardt LF . Lack of neurotrophin-3 results in death of spinal sensory neurons and premature differentiation of their precursors. Neuron 1996; 17: 1065–1078.
Reubinoff BE et al. Neural progenitors from human embryonic stem cells. Nat Biotechnol 2001; 19: 1134–1140.
Turgeman G et al. Engineered human mesenchymal stem cells: a novel platform for skeletal cell mediated gene therapy. J Gene Med 2001; 3: 240–251.
Pittenger MF et al. Multilineage potential of adult human mesenchymal stem cells. Science 1999; 284: 143–147.
Caplan AI . Mesenchymal stem cells. J Orthop Res 1991; 9: 641–650.
Bruder SP, Fink DJ, Caplan AI . Mesenchymal stem cells in bone development, bone repair, and skeletal regeneration therapy. J Cell Biochem 1994; 56: 283–294.
Lennon DP et al. Dilution of human mesenchymal stem cells with dermal fibroblasts and the effects on in vitro and in vivo osteochondrogenesis. Dev Dyn 2000; 219: 50–62.
Jacobs JP, Jones CM, Baille JP . Characteristics of a human diploid cell designated MRC-5. Nature 1970; 227: 168–170.
Yotnda P et al. Efficient infection of primitive hematopoietic stem cells by modified adenovirus. Gene Therapy 2001; 8: 930–937.
Shayakhmetov DM, Papayannopoulou T, Stamatoyannopoulos G, Lieber A . Efficient gene transfer into human CD34(+) cells by a retargeted adenovirus vector. J Virol 2000; 74: 2567–2583.
Acknowledgements
We thank Dr Malcolm Brenner for his insightful review of the manuscript and Dr Robert McAlhany for virus preparation. We also thank Genetics Institute for the anti-BMP2 monoclonal antibody (h3b2/17.8.1). This work was supported by National Institutes of Health Grants R03AR47463-01 (EAO-D) and R21AR484-01 (ARD).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gugala, Z., Olmsted-Davis, E., Gannon, F. et al. Osteoinduction by ex vivo adenovirus-mediated BMP2 delivery is independent of cell type. Gene Ther 10, 1289–1296 (2003). https://doi.org/10.1038/sj.gt.3302006
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302006
Keywords
This article is cited by
-
Orthopedic Gene Therapy in 2008
Molecular Therapy (2009)
-
Comparison of Osteogenic Potentials of BMP4 Transduced Stem Cells from Autologous Bone Marrow and Fat Tissue in a Rabbit Model of Calvarial Defects
Calcified Tissue International (2009)
-
Ex vivo-transduced autologous skin fibroblasts expressing human Lim mineralization protein-3 efficiently form new bone in animal models
Gene Therapy (2008)
-
Ectopic bone formation in vivo induced by a novel synthetic peptide derived from BMP-2 using porous collagen scaffolds
Journal of Wuhan University of Technology-Mater. Sci. Ed. (2007)
-
Endochondral Bone Formation from Hydrogel Carriers Loaded with BMP2-transduced Cells
Annals of Biomedical Engineering (2007)