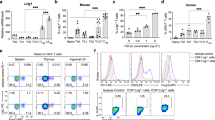
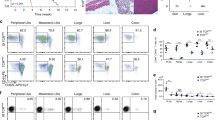
Abstract
The ability to produce vigorous immune responses that spare self tissues and organs depends on the elimination of autoreactive T and B cells. However, purging of immature and mature self-reactive T and B cells is incomplete and may also require the involvement of cells programmed to suppress immune responses1. Regulatory T cells (Treg) belonging to the CD4+ T-cell subset may have a role in preventing untoward inflammatory responses, but T-cell subsets programmed to inhibit the development of autoantibody formation and systemic-lupus-erythematosus-like disease have not yet been defined2. Here we delineate a CD8+ regulatory T-cell lineage that is essential for the maintenance of self tolerance and prevention of murine autoimmune disease. Genetic disruption of the inhibitory interaction between these CD8+ T cells and their target Qa-1+ follicular T-helper cells results in the development of a lethal systemic-lupus-erythematosus-like autoimmune disease. These findings define a sublineage of CD8 T cells programmed to suppress rather than activate immunity that represents an essential regulatory element of the immune response and a guarantor of self tolerance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cantor, H. T-cell receptor crossreactivity and autoimmune disease. Adv. Immunol. 75, 209–233 (2000)
Cantor, H. Reviving suppression? Nature Immunol. 5, 347–349 (2004)
Josefowicz, S. Z. & Rudensky, A. Control of regulatory T cell lineage commitment and maintenance. Immunity 30, 616–625 (2009)
Noble, A., Zhao, Z.-S. & Cantor, H. Suppression of immune responses by CD8 cells: II. Qa-1 on activated B-cells stimulates CD8 cell suppression of T helper 2 responses. J. Immunol. 160, 566–571 (1998)
Jiang, H., Zhang, S.-L. & Pernis, B. Role of CD8+ T cells in murine experimental allergic encephalomyelitis. Science 256, 1213–1215 (1992)
Hu, D. et al. Analysis of regulatory CD8 T cells in Qa-1-deficient mice. Nature Immunol. 5, 516–523 (2004)
Chen, W. et al. Perceiving the avidity of T cell activation can be translated into peripheral T cell regulation. Proc. Natl Acad. Sci. USA 104, 20472–20477 (2007)
Lu, L. & Cantor, H. Generation and regulation of CD8+ regulatory T cells. Cell Mol. Immunol. 5, 401–406 (2008)
Lu, L., Kim, H. J., Werneck, M. B. & Cantor, H. Regulation of CD8+ regulatory T cells: Interruption of the NKG2A-Qa-1 interaction allows robust suppressive activity and resolution of autoimmune disease. Proc. Natl Acad. Sci. USA 105, 19420–19425 (2008)
Lu, L. et al. Regulation of activated CD4+ T cells by NK cells via the Qa-1–NKG2A inhibitory pathway. Immunity 26, 593–604 (2007)
Bubier, J. A. et al. A critical role for IL-21 receptor signaling in the pathogenesis of systemic lupus erythematosus in BXSB-Yaa mice. Proc. Natl Acad. Sci. USA 106, 1518–1523 (2009)
Linterman, M. A. et al. Follicular helper T cells are required for systemic autoimmunity. J. Exp. Med. 206, 561–576 (2009)
Sangster, M. Y. et al. Analysis of the virus-specific and nonspecific B cell response to a persistent B-lymphotropic gammaherpesvirus. J. Immunol. 164, 1820–1828 (2000)
Panoutsakopoulou, V. et al. Analysis of the relationship between viral infection and autoimmune disease. Immunity 15, 137–147 (2001)
Hunziker, L. et al. Hypergammaglobulinemia and autoantibody induction mechanisms in viral infections. Nature Immunol. 4, 343–349 (2003)
Mitchell, D. M., Ravkov, E. V. & Williams, M. A. Distinct roles for IL-2 and IL-15 in the differentiation and survival of CD8+ effector and memory T cells. J. Immunol. 184, 6719–6730 (2010)
Zeng, R. et al. Synergy of IL-21 and IL-15 in regulating CD8+ T cell expansion and function. J. Exp. Med. 201, 139–148 (2005)
Nurieva, R. I. et al. Generation of T follicular helper cells is mediated by interleukin-21 but independent of T helper 1, 2, or 17 cell lineages. Immunity 29, 138–149 (2008)
Wardemann, H. et al. Predominant autoantibody production by early human B cell precursors. Science 301, 1374–1377 (2003)
Yurasov, S. & Nussenzweig, M. C. Regulation of autoreactive antibodies. Curr. Opin. Rheumatol. 19, 421–426 (2007)
Hao, Z. et al. Fas receptor expression in germinal-center B cells is essential for T and B lymphocyte homeostasis. Immunity 29, 615–627 (2008)
Ray, S. K., Putterman, C. & Diamond, B. Pathogenic autoantibodies are routinely generated during the response to foreign antigen: a paradigm for autoimmune disease. Proc. Natl Acad. Sci. USA 93, 2019–2024 (1996)
Simpson, N. et al. Expansion of circulating T cells resembling follicular helper T cells is a fixed phenotype that identifies a subset of severe systemic lupus erythematosus. Arthritis Rheum. 62, 234–244 (2010)
Acknowledgements
We thank R. Bronson (DF/HCC Rodent Histopathology Core) for histology analysis, R. Gelman and J. Yang for help with statistical analyses, C. Daniel and T. Kreslavsky for cytometry support and critical discussions, M. Iannacone and U. von Andrian for LCMV, X. Wang for technical assistance, M. Call, D. A. Alvarez Arias, T. Kadakia and A. Angel for manuscript and figure preparation. This work was supported in part by NIH Research Grant AI 037562, the Lupus Research Institute and a gift from the Schecter Research Foundation to H.C.; National Research Service Award Fellowships (DFCI/NCI T32 CA070083) to H.-J.K. and (HSPH/NCI T32 CA009382) to X.T.; and a Belgian American Educational Foundation Fellowship to B.V.
Author information
Authors and Affiliations
Contributions
H.-J.K. and H.C. conceived and planned experiments; H.-J.K., B.V., X.T. and L.L. performed experiments; H.-J.K. and H.C. analysed data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-17 with legends. (PDF 1134 kb)
Rights and permissions
About this article
Cite this article
Kim, HJ., Verbinnen, B., Tang, X. et al. Inhibition of follicular T-helper cells by CD8+ regulatory T cells is essential for self tolerance. Nature 467, 328–332 (2010). https://doi.org/10.1038/nature09370
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature09370
This article is cited by
-
The endogenous repertoire harbors self-reactive CD4+ T cell clones that adopt a follicular helper T cell-like phenotype at steady state
Nature Immunology (2023)
-
TGFβ control of immune responses in cancer: a holistic immuno-oncology perspective
Nature Reviews Immunology (2023)
-
The regulation of Tfh cell differentiation by β-hydroxybutyrylation modification of transcription factor Bcl6
Chromosoma (2023)
-
High expression of OX-40, ICOS, and low expression PD-L1 of follicular helper and follicular cytotoxic T cells in chronic lymphocytic leukemia
Journal of Hematopathology (2022)
-
Altered levels of circulating CD8+CXCR5+PD-1+T follicular cytotoxic cells in primary Sjögren’s syndrome
Clinical Rheumatology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.