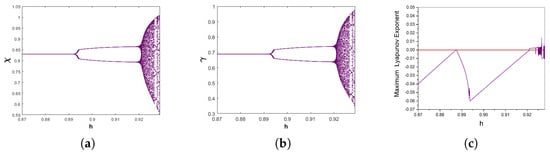
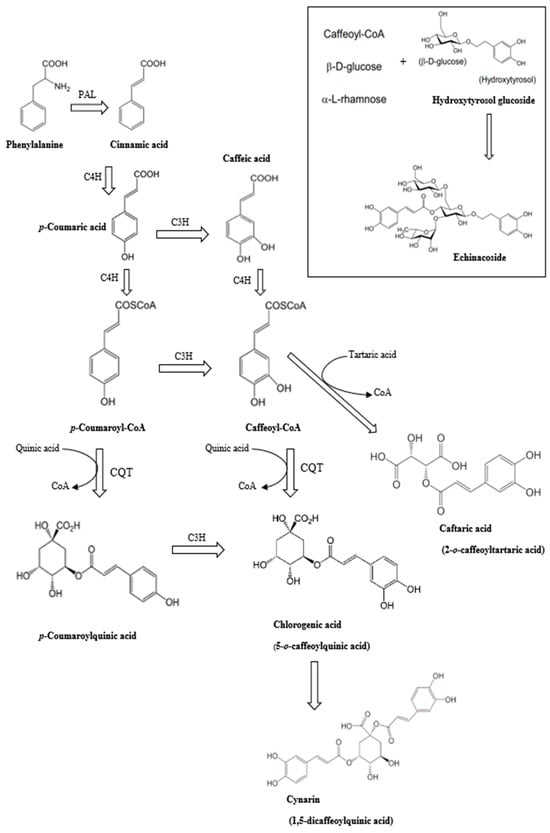
Salmonella is a common foodborne pathogen that can cause food poisoning, posing a serious threat to human health. Therefore, quickly, sensitively, and accurately detecting
Salmonella is crucial to ensuring food safety. For the
Salmonella hilA gene, we designed Recombinase-aided amplification (RAA) primers
[...] Read more.
Salmonella is a common foodborne pathogen that can cause food poisoning, posing a serious threat to human health. Therefore, quickly, sensitively, and accurately detecting
Salmonella is crucial to ensuring food safety. For the
Salmonella hilA gene, we designed Recombinase-aided amplification (RAA) primers and dsDNA-specific nuclease (DNase) probes. The ideal primer and probe combination was found when conditions were optimized. Under UV light, a visual
Salmonella detection technique (RAA-dsDNase) was developed. Additionally, the RAA-dsDNase was modified to further reduce pollution hazards and simplify operations. One-pot RAA-dsDNase-UV or one-pot RAA-dsDNase-LFD was developed as a
Salmonella detection method, using UV or a lateral flow dipstick (LFD) for result observation. Among them, one-pot RAA-dsDNase and one-pot RAA-dsDNase-LFD had detection times of 50 min and 60 min, respectively, for detecting
Salmonella genomic DNA. One-pot RAA-dsDNase-UV had a detection limit of 10
1 copies/μL and 10
1 CFU/mL, while one-pot RAA-dsDNase-LFD had a sensitivity of 10
2 copies/μL and 10
2 CFU/mL. One-pot RAA-dsDNase-UV and one-pot RAA-dsDNase-LFD assays may identify 17 specific
Salmonella serovars witho ut causing a cross-reaction with the remaining 8 bacteria, which include
E. coli. Furthermore,
Salmonella in tissue and milk samples has been reliably detected using both approaches. Overall, the detection method developed in this study can quickly, sensitively, and accurately detect
Salmonella, and it is expected to become an important detection tool for the prevention and control of
Salmonella in the future.
Full article
 IJMS
IMPACT
IJMS
IMPACT Applied Sciences
IMPACT
Applied Sciences
IMPACT Sustainability
IMPACT
Sustainability
IMPACT Sensors
IMPACT
Sensors
IMPACT JCM
IMPACT
JCM
IMPACT Energies
IMPACT
Energies
IMPACT Molecules
IMPACT
Molecules
IMPACT Materials
IMPACT
Materials
IMPACT Remote Sensing
IMPACT
Remote Sensing
IMPACT Cancers
IMPACT
Cancers
IMPACT Electronics
IMPACT
Electronics
IMPACT Mathematics
IMPACT
Mathematics
IMPACT Foods
IMPACT
Foods
IMPACT Buildings
IMPACT
Buildings
IMPACT Plants
IMPACT
Plants
IMPACT Nutrients
IMPACT
Nutrients
IMPACT Animals
IMPACT
Animals
IMPACT Polymers
IMPACT
Polymers
IMPACT Water
IMPACT
Water
IMPACT Diagnostics
IMPACT
Diagnostics
IMPACT Biomedicines
IMPACT
Biomedicines
IMPACT Agronomy
IMPACT
Agronomy
IMPACT Microorganisms
IMPACT
Microorganisms
IMPACT Processes
IMPACT
Processes
IMPACT Healthcare
IMPACT
Healthcare
IMPACT Forests
IMPACT
Forests
IMPACT Cells
IMPACT
Cells
IMPACT JMSE
IMPACT
JMSE
IMPACT Medicina
IMPACT
Medicina
IMPACT Viruses
IMPACT
Viruses
IMPACT Agriculture
IMPACT
Agriculture
IMPACT Nanomaterials
IMPACT
Nanomaterials
IMPACT IJERPH
IJERPH
 Land
IMPACT
Land
IMPACT Pharmaceutics
IMPACT
Pharmaceutics
IMPACT Pharmaceuticals
IMPACT
Pharmaceuticals
IMPACT Religions
IMPACT
Religions
IMPACT Biomolecules
IMPACT
Biomolecules
IMPACT Life
IMPACT
Life
IMPACT Micromachines
IMPACT
Micromachines
IMPACT Atmosphere
IMPACT
Atmosphere
IMPACT Antioxidants
IMPACT
Antioxidants
IMPACT Genes
IMPACT
Genes
IMPACT Metals
IMPACT
Metals
IMPACT Symmetry
IMPACT
Symmetry
IMPACT Children
IMPACT
Children
IMPACT Coatings
IMPACT
Coatings
IMPACT Vaccines
IMPACT
Vaccines
IMPACT Horticulturae
IMPACT
Horticulturae
IMPACT Education Sciences
IMPACT
Education Sciences
IMPACT Minerals
IMPACT
Minerals
IMPACT Brain Sciences
IMPACT
Brain Sciences
IMPACT JPM
IMPACT
JPM
IMPACT Bioengineering
IMPACT
Bioengineering
IMPACT