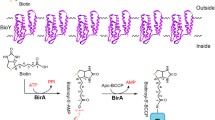
Abstract
The biosynthesis of the pimelate moiety of biotin in Escherichia coli requires two specialized proteins, BioC and BioH. However, the enzymes that have BioC- or BioH-like activities show remarkable sequence diversity among biotin-producing bacteria. Here, we report that the intracellular rickettsial pathogen Ehrlichia chaffeensis encodes two novel proteins, BioT and BioU, which functionally replace the E. coli BioC and BioH proteins, respectively. The desthiobiotin assays demonstrated that these two proteins make pimeloyl-acyl carrier protein (ACP) from the substrate malonyl-ACP with the aid of the FAS II pathway, through the expected pimeloyl-ACP methyl ester intermediate. BioT and BioU homologues seem restricted to the species of Ehrlichia and its close relative, Anaplasma. Taken together, the synthesis of the biotin precursor in E. chaffeensis appears to be catalyzed by two novel BioC- and BioH-like proteins.





Similar content being viewed by others
References
Attwood PV, Wallace JC (2002) Chemical and catalytic mechanisms of carboxyl transfer reactions in biotin-dependent enzymes. ACC Chem Res 35(2):113–120
Finkenwirth F, Kirsch F, Eitinger T (2014) A versatile Escherichia coli strain for identification of biotin transporters and for biotin quantification. Bioengineered 5(2):129–132. https://doi.org/10.4161/bioe.26887
Cronan JE Jr (2001) The biotinyl domain of Escherichia coli acetyl-CoA carboxylase. Evidence that the “thumb” structure id essential and that the domain functions as a dimer. J Biol Chem 276(40):37355–37364. https://doi.org/10.1074/jbc.M106353200
Cronan JE (2014) Biotin and lipoic acid: synthesis, attachment, and regulation. EcoSal Plus. https://doi.org/10.1128/ecosalplus.ESP-0001-2012
Lin S, Cronan JE (2012) The BioC O-methyltransferase catalyzes methyl esterification of malonyl-acyl carrier protein, an essential step in biotin synthesis. J Biol Chem 287(44):37010–37020. https://doi.org/10.1074/jbc.M112.410290
Agarwal V, Lin S, Lukk T, Nair SK, Cronan JE (2012) Structure of the enzyme-acyl carrier protein (ACP) substrate gatekeeper complex required for biotin synthesis. Proc Natl Acad Sci USA 109(43):17406–17411. https://doi.org/10.1073/pnas.1207028109
Lin S, Cronan JE (2011) Closing in on complete pathways of biotin biosynthesis. Mol Biosyst 7(6):1811–1821. https://doi.org/10.1039/c1mb05022b
Lin S, Hanson RE, Cronan JE (2010) Biotin synthesis begins by hijacking the fatty acid synthetic pathway. Nat Chem Biol 6(9):682–688. https://doi.org/10.1038/nchembio.420
Rodionov DA, Mironov AA, Gelfand MS (2002) Conservation of the biotin regulon and the BirA regulatory signal in Eubacteria and Archaea. Genome Res 12(10):1507–1516. https://doi.org/10.1101/gr.314502
Shapiro MM, Chakravartty V, Cronan JE (2012) Remarkable diversity in the enzymes catalyzing the last step in synthesis of the pimelate moiety of biotin. PLoS ONE 7(11):e49440. https://doi.org/10.1371/journal.pone.0049440
Feng Y, Napier BA, Manandhar M, Henke SK, Weiss DS, Cronan JE (2014) A Francisella virulence factor catalyses an essential reaction of biotin synthesis. Mol Microbiol 91(2):300–314. https://doi.org/10.1111/mmi.12460
Bi H, Zhu L, Jia J, Cronan JE (2016) A biotin biosynthesis gene restricted to helicobacter. Sci Rep 6:21162. https://doi.org/10.1038/srep21162
Polgar L (2005) The catalytic triad of serine peptidases. Cell Mol Life Sci 62(19–20):2161–2172. https://doi.org/10.1007/s00018-005-5160-x
Estrada P, Manandhar M, Dong SH, Deveryshetty J, Agarwal V, Cronan JE, Nair SK (2017) The pimeloyl-CoA synthetase BioW defines a new fold for adenylate-forming enzymes. Nat Chem Biol 13(6):668–674. https://doi.org/10.1038/nchembio.2359
Manandhar M, Cronan JE (2017) Pimelic acid, the first precursor of the Bacillus subtilis biotin synthesis pathway, exists as the free acid and is assembled by fatty acid synthesis. Mol Microbiol 104(4):595–607. https://doi.org/10.1111/mmi.13648
Entcheva P, Phillips DA, Streit WR (2002) Functional analysis of Sinorhizobium meliloti genes involved in biotin synthesis and transport. Appl Environ Microbiol 68(6):2843–2848
Dumler JS, Barbet AF, Bekker CP, Dasch GA, Palmer GH, Ray SC, Rikihisa Y, Rurangirwa FR (2001) Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and ‘HGE agent’ as subjective synonyms of Ehrlichia phagocytophila. Int J Syst Evol Microbiol 51(Pt 6):2145–2165. https://doi.org/10.1099/00207713-51-6-2145
Rikihisa Y (2015) Molecular pathogenesis of Ehrlichia chaffeensis infection. Annu Rev Microbiol 69:283–304. https://doi.org/10.1146/annurev-micro-091014-104411
Telford SR III, Dawson JE, Katavolos P, Warner CK, Kolbert CP, Persing DH (1996) Perpetuation of the agent of human granulocytic ehrlichiosis in a deer tick-rodent cycle. Proc Natl Acad Sci USA 93(12):6209–6214
Dumler JS, Choi KS, Garcia-Garcia JC, Barat NS, Scorpio DG, Garyu JW, Grab DJ, Bakken JS (2005) Human granulocytic anaplasmosis and Anaplasma phagocytophilum. Emerg Infect Dis 11(12):1828–1834. https://doi.org/10.3201/eid1112.050898
Paddock CD, Childs JE (2003) Ehrlichia chaffeensis: a prototypical emerging pathogen. Clin Microbiol Rev 16(1):37–64
Woong Park S, Klotzsche M, Wilson DJ, Boshoff HI, Eoh H, Manjunatha U, Blumenthal A, Rhee K, Barry CE III, Aldrich CC, Ehrt S, Schnappinger D (2011) Evaluating the sensitivity of Mycobacterium tuberculosis to biotin deprivation using regulated gene expression. PLoS Pathog 7(9):e1002264. https://doi.org/10.1371/journal.ppat.1002264
Napier BA, Meyer L, Bina JE, Miller MA, Sjostedt A, Weiss DS (2012) Link between intraphagosomal biotin and rapid phagosomal escape in Francisella. Proc Natl Acad Sci USA 109(44):18084–18089. https://doi.org/10.1073/pnas.1206411109
Zlitni S, Ferruccio LF, Brown ED (2013) Metabolic suppression identifies new antibacterial inhibitors under nutrient limitation. Nat Chem Biol 9(12):796–804. https://doi.org/10.1038/nchembio.1361
Park SW, Casalena DE, Wilson DJ, Dai R, Nag PP, Liu F, Boyce JP, Bittker JA, Schreiber SL, Finzel BC, Schnappinger D, Aldrich CC (2015) Target-based identification of whole-cell active inhibitors of biotin biosynthesis in Mycobacterium tuberculosis. Chem Biol 22(1):76–86. https://doi.org/10.1016/j.chembiol.2014.11.012
Dunning Hotopp JC, Lin M, Madupu R, Crabtree J, Angiuoli SV, Eisen JA, Seshadri R, Ren Q, Wu M, Utterback TR, Smith S, Lewis M, Khouri H, Zhang C, Niu H, Lin Q, Ohashi N, Zhi N, Nelson W, Brinkac LM, Dodson RJ, Rosovitz MJ, Sundaram J, Daugherty SC, Davidsen T, Durkin AS, Gwinn M, Haft DH, Selengut JD, Sullivan SA, Zafar N, Zhou L, Benahmed F, Forberger H, Halpin R, Mulligan S, Robinson J, White O, Rikihisa Y, Tettelin H (2006) Comparative genomics of emerging human ehrlichiosis agents. PLoS Genet 2(2):e21. https://doi.org/10.1371/journal.pgen.0020021
Choi-Rhee E, Cronan JE (2005) A nucleosidase required for in vivo function of the S-adenosyl-L-methionine radical enzyme, biotin synthase. Chem Biol 12(5):589–593. https://doi.org/10.1016/j.chembiol.2005.04.012
Khan SR, Gaines J, Roop RM II, Farrand SK (2008) Broad-host-range expression vectors with tightly regulated promoters and their use to examine the influence of TraR and TraM expression on Ti plasmid quorum sensing. Appl Environ Microbiol 74(16):5053–5062. https://doi.org/10.1128/AEM.01098-08
Guzman LM, Belin D, Carson MJ, Beckwith J (1995) Tight regulation, modulation, and high-level expression by vectors containing the arabinose PBAD promoter. J Bacteriol 177(14):4121–4130
Zhu L, Lin J, Ma J, Cronan JE, Wang H (2010) Triclosan resistance of Pseudomonas aeruginosa PAO1 is due to FabV, a triclosan-resistant enoyl-acyl carrier protein reductase. Antimicrob Agents Chemother 54(2):689–698. https://doi.org/10.1128/AAC.01152-09
Jiang Y, Chan CH, Cronan JE (2006) The soluble acyl-acyl carrier protein synthetase of Vibrio harveyi B392 is a member of the medium chain acyl-CoA synthetase family. Biochemistry 45(33):10008–10019. https://doi.org/10.1021/bi060842w
Bi H, Yu Y, Dong H, Wang H, Cronan JE (2014) Xanthomonas campestris RpfB is a fatty Acyl-CoA ligase required to counteract the thioesterase activity of the RpfF diffusible signal factor (DSF) synthase. Mol Microbiol 93(2):262–275. https://doi.org/10.1111/mmi.12657
Cronan JE, Thomas J (2009) Bacterial fatty acid synthesis and its relationships with polyketide synthetic pathways. Methods Enzymol 459:395–433. https://doi.org/10.1016/S0076-6879(09)04617-5
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33(7):1870–1874. https://doi.org/10.1093/molbev/msw054
Ismail N, McBride JW (2017) Tick-borne emerging infections: Ehrlichiosis and Anaplasmosis. Clin Lab Med 37(2):317–340. https://doi.org/10.1016/j.cll.2017.01.006
Finn RD, Coggill P, Eberhardt RY, Eddy SR, Mistry J, Mitchell AL, Potter SC, Punta M, Qureshi M, Sangrador-Vegas A, Salazar GA, Tate J, Bateman A (2016) The Pfam protein families database: towards a more sustainable future. Nucleic Acids Res 44(D1):D279–D285. https://doi.org/10.1093/nar/gkv1344
Shi J, Cao X, Chen Y, Cronan JE, Guo Z (2016) An Atypical alpha/beta-hydrolase fold revealed in the crystal structure of pimeloyl-acyl carrier protein methyl esterase BioG from Haemophilus influenzae. Biochemistry 55(48):6705–6717. https://doi.org/10.1021/acs.biochem.6b00818
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant Nos. 31600039 and 31570053). We thank Dr. Xue-Jie Yu for providing genomic DNA samples.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hang, X., Zeng, Q., Zeng, L. et al. Functional Replacement of the BioC and BioH Proteins of Escherichia coli Biotin Precursor Biosynthesis by Ehrlichia chaffeensis Novel Proteins. Curr Microbiol 76, 626–636 (2019). https://doi.org/10.1007/s00284-019-01669-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00284-019-01669-w