Abstract
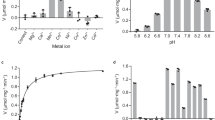
LACTATE dehydrogenase (L-lactate: NAD oxidoreductase, EC 1.1.1.27) occurs in most animal tissues in five different forms1,2, which can easily be separated by electrophoresis, and these dehydrogenases are enzymes of substrate specificity. Meister3, however, has found that a series of α,γ-diketo acids are reduced at an essentially uniform rate which is about one-tenth that for pyruvate. Markert and Møller1 observed that a series of α-hydroxy acids are oxidized at different rates by the isozymes of bovine heart extract, and Kun4 has shown that β-mer-captopyruvate is reduced by crystalline heart lactate dehydrogenase. It has been shown (unpublished) that β-hydroxybutyrate is a slowly reacting substrate. The present report presents evidence for the glyoxylate reductase activity (EC 1.1.1.26) of lactate dehydrogenase based on quantitative and qualitative studies of purified enzyme (Boehringer rabbit muscle lactate dehydrogenase) or rat liver extract. Quantitative determination of dehydrogenase activities was carried out by measurement of the diminution or production of reduced nicotinamide dinucleotide (NADH2) (at 23° C and at a wave-length of 340 mµ).
Similar content being viewed by others
Article PDF
References
Markert, C. L., and Møller, F., Proc. U.S. Nat. Acad. Sci., 45, 753 (1959).
Wieland, T., and Pfleiderer, G., Biochem. Z., 329, 112 (1957).
Meister, A., J. Biol. Chem., 184, 117 (1950).
Kun, E., Biochim. Biophys. Acta, 25, 135 (1957).
Sawaki, S., and Morikawa, N., J. Vitaminol., 10, 179 (1964).
Kun, E., J. Biol. Chem., 194, 601 (1952).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
SAWAKI, S., YAMADA, K. Glyoxylate Reductase Activity of Lactate Dehydrogenase. Nature 210, 91 (1966). https://doi.org/10.1038/210091a0
Issue Date:
DOI: https://doi.org/10.1038/210091a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.