Abstract
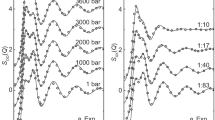
THE variation with pressure of the dielectric constant of the alkali halides has been measured, for selected salts, by Mayburg1 and Jarman2, whose work is summarized in Table 1. Jarman deduced that the polarizability of the unit cell, defined below, was proportional to the square of the volume; although lithium fluoride and magnesium oxide appear to be exceptions to this rule.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Mayburg, G., Phys. Rev., 79, 375 (1950).
Jarman, G. M., Ph.D. thesis, University of Bristol (1957).
Thomson, A. M., Inst. Rad. Eng. Proc., “Instrumentation” (Dec. 1958).
The Clausius–Mosotti Relation; for a discussion of which see Fröhlich, H., Theory of Dielectrics (Oxford Univ. Press, 1949).
Höjendahl, K., K. Dansk Vidensk. Selskab, 16, No. 2 (1938).
Bridgman, P. W., Proc. Amer. Acad. Arts and Sci., 76, 1 (1945).
Pauling, L., quoted in Kittell, C., Introduction to Solid State Physics, second ed., 67 (J. Wiley, 1956).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
HILL, G. Variation of the Dielectric Constant of the Alkali Halides with Pressure. Nature 193, 1275–1276 (1962). https://doi.org/10.1038/1931275a0
Issue Date:
DOI: https://doi.org/10.1038/1931275a0
This article is cited by
-
Infrared spectra of small interstellar grains
Astrophysics and Space Science (1976)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.