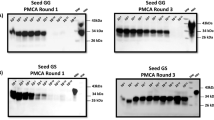
Abstract
There is increasing concern over the extent to which bovine spongiform encephalopathy (BSE) prions have been transmitted to humans, as a result of the rising number of variant Creutzfeldt–Jakob disease (vCJD) cases. Toward preventing new transmissions, diagnostic tests for prions in livestock have been developed using the conformation-dependent immunoassay (CDI), which simultaneously measures specific antibody binding to denatured and native forms of the prion protein (PrP). We employed high-affinity recombinant antibody fragments (recFab) reacting with residues 95–105 of bovine (Bo) PrP for detection and another recFab that recognizes residues 132–156 for capture in the CDI. We report that the CDI is capable of measuring the disease-causing PrP isoform (PrPSc) in bovine brainstems with a sensitivity similar to that of end-point titrations in transgenic (Tg) mice expressing BoPrP. Prion titers were ∼107 ID50 units per gram of bovine brainstem when measured in Tg(BoPrP) mice, a figure ∼10 times greater than that determined by bioassay in cattle and ∼10,000× greater than in wild-type mice. We also report substantial differences in BoPrPSc levels in different areas of the obex region, where neuropathology has been consistently observed in cattle with BSE. The CDI was able to discriminate between PrPSc from BSE-infected cattle and Tg(BoPrP) mice as well as from chronic wasting disease (CWD)-infected deer and elk. Our findings argue that applying the CDI to livestock should considerably reduce human exposure to animal prions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mehlhorn, I. et al. High-level expression and characterization of a purified 142-residue polypeptide of the prion protein. Biochemistry 35, 5528–5537 (1996).
Safar, J. et al. Eight prion strains have PrPSc molecules with different conformations. Nat. Med. 4, 1157–1165 (1998).
Bruce, M. et al. Transmission of bovine spongiform encephalopathy and scrapie to mice: strain variation and the species barrier. Phil. Trans. R. Soc. Lond. B 343, 405–411 (1994).
Fraser, H., Bruce, M.E., Chree, A., McConnell, I. & Wells, G.A.H. Transmission of bovine spongiform encephalopathy and scrapie to mice. J. Gen. Virol. 73, 1891–1897 (1992).
Moynagh, J., Schimmel, H. & Kramer, G.N. The evaluation of tests for the diagnosis of transmissible spongiform encephalopathy in bovines. 1–36 (European Commission, 1999).
Deslys, J.P. et al. Screening slaughtered cattle for BSE. Nature 409, 476–478 (2001).
Wells, G.A.H. et al. Preliminary observations on the pathogenesis of experimental bovine spongiform encephalopathy (BSE): an update. Vet. Rec. 142, 103–106 (1998).
Scott, M.R. et al. Compelling transgenetic evidence for transmission of bovine spongiform encephalopathy prions to humans. Proc. Natl. Acad. Sci. USA 96, 15137–15142 (1999).
Peretz, D. et al. Strain-specified relative conformational stability of the scrapie prion protein. Protein Sci. 10, 854–863 (2001).
Schaller, O. et al. Validation of a western immunoblotting procedure for bovine PrPSc detection and its use as a rapid surveillance method for the diagnosis of bovine spongiform encephalopathy (BSE). Acta Neuropathol. 98, 437–443 (1999).
Biffiger, K. et al. Validation of a luminescence immunoassay for the detection of PrPSc in brain homogenate. J. Virol. Methods 101, 79–84 (2002).
Bosque, P.J. et al. Prions in skeletal muscle. Proc. Natl. Acad. Sci. USA 99, 3812–3817 (2002).
Prusiner, S.B. & Safar, J.G. Method of concentrating prion proteins in blood samples. US 6,166,187 (2000).
Williamson, R.A. et al. Circumventing tolerance to generate autologous monoclonal antibodies to the prion protein. Proc. Natl. Acad. Sci. USA 93, 7279–7282 (1996).
Peretz, D. et al. A conformational transition at the N terminus of the prion protein features in formation of the scrapie isoform. J. Mol. Biol. 273, 614–622 (1997).
Peretz, D. et al. Antibodies inhibit prion propagation and clear cell cultures of prion infectivity. Nature 412, 739–743 (2001).
Burton, D.R. & Barbas, C.F. Human antibodies from combinatorial libraries. Adv. Immunol. 57, 191–280 (1994).
Johnsson, B., Lofas, S. & Lindquist, G. Immobilization of proteins to a carboxymethyldextran-modified gold surface for biospecific interaction analysis in surface plasmon resonance sensors. Anal. Biochem. 198, 268–277 (1991).
Dougherty, R. in Techniques in Experimental Virology (ed. Harris, R.J.C.) 169–224 (Academic Press, New York, 1964).
Prusiner, S.B. et al. Immunologic and molecular biological studies of prion proteins in bovine spongiform encephalopathy. J. Infect. Dis. 167, 602–613 (1993).
Wells, G.A.H. & Wilesmith, J.W. The neuropathology and epidemiology of bovine spongiform encephalopathy. Brain Pathol. 5, 91–103 (1995).
Acknowledgements
The authors thank Elizabeth Williams (Wyoming State Veterinary Laboratory, University of Wyoming, Laramie) for providing the CWD samples, Hank Baron and Fred Feldman for encouragement and stimulating discussions, and the British Ministry of Agriculture, Fisheries and Food (MAFF) and VLA (UK) for supplying BSE brainstems. This work was supported by grants from the National Institutes of Health (AG02132, AG010770, NS22786, and NS14069) and MAFF (ES1756), as well as by gifts from the G. Harold and Leila Y. Mathers Charitable Foundation and from Centeon.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
J.G.S., J.M., G.L, H.S., D.G., and S.B.P. hold stock shares in InPro Biotechnology, which holds an exclusive license from the University of Calfornia for the conformation-dependent immunoassay (CDI).
Supplementary information
Rights and permissions
About this article
Cite this article
Safar, J., Scott, M., Monaghan, J. et al. Measuring prions causing bovine spongiform encephalopathy or chronic wasting disease by immunoassays and transgenic mice. Nat Biotechnol 20, 1147–1150 (2002). https://doi.org/10.1038/nbt748
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt748
This article is cited by
-
Evolving prion-like tau conformers differentially alter postsynaptic proteins in neurons inoculated with distinct isolates of Alzheimer’s disease tau
Cell & Bioscience (2023)
-
New developments in prion disease research using genetically modified mouse models
Cell and Tissue Research (2023)
-
Emergence of CWD strains
Cell and Tissue Research (2023)
-
Diverse, evolving conformer populations drive distinct phenotypes in frontotemporal lobar degeneration caused by the same MAPT-P301L mutation
Acta Neuropathologica (2020)
-
Artificial strain of human prions created in vitro
Nature Communications (2018)