Abstract
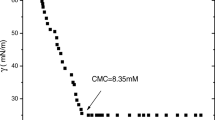
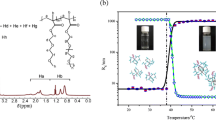
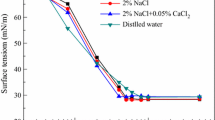
A solvated and cross-linked copolymer of N-isopropylacrylamide (IPAAm) and 2-(acrylamido)-2-methyl propane sulfonic acid (AMPS) was synthesized and its interaction with cationic surfactant lauryl-pyridinium chloride (C12PyCl) was investigated. The solvated copolymer exhibited a lower critical solution temperature (LCST) in water, which was extensively shifted to a higher temperature due to the increase of hydrophilicity introduced by AMPS. In C12PyCl solution, LCST of the copolymer was dramatically decreased due to the binding of C12PyCl to AMPS unit, forming a stoichiometric complex. However, in the concentrated C12PyCl solution, its LCST increased due to the non-stoichiometric complex formation. This phenomenon was further examined in the cross-linked copolymer, analyzed by binding isotherms. Two-step binding of surfactant was demonstrated followed by gel shrinking and re-swelling. This binding mechanism was further discussed regarding the effect of charge density and the hydro-phobicity of the main-chain backbone in terms of electrostatic and hydrophobic interactions.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 13 May 1997 Accepted: 13 August 1997
Rights and permissions
About this article
Cite this article
Matsukata, M., Hirata, M., Gong, J. et al. Two-step surfactant binding of solvated and cross-linked poly(N-isopropylacrylamide-co- (2-acrylamido-2-methyl propane sulfonic acid)). Colloid Polym Sci 276, 11–18 (1998). https://doi.org/10.1007/s003960050202
Issue Date:
DOI: https://doi.org/10.1007/s003960050202