Abstract
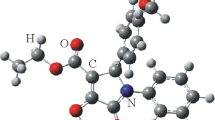
Heating of 2-benzylidene-1-indanone (1) either in dimethylformamide in the presence of guanidine carbonate or inn-propanol in the presence of thiourea and sodium propylate yields a dimerP with the structure of 1,3-diphenyl-3a,8a-dihydrospiro{cyclopenta[a]indene-2,2′(1H,3′H)-indene}-1′,8(3H)-dione (7); this was deduced from the 400 MHz-1H- and 100MHz-13C-nmr-spectra and the corresponding two-dimensional HH- and CH-correlations. A possible mechanism for the formation of the spirocompound 7 is proposed; the stereo formula of the dimer7 K was established on the basis of a 400 MHz-HH-NOESY experiment.
Similar content being viewed by others
Literatur
Wendelin W, Schermanz K, Kerbl J (1983) Monatsh Chem 114: 717
Wendelin W, Kerbl J (1984) Monatsh Chem 115: 309
Wendelin W, Schermanz K (1984) J Heterocycl Chem 21: 65
Willems J,Vandenberghe A (1960) Chem Abstr 54: 22657b
Zigeuner G, Gübitz G, Eisenreich V (1970) Monatsh Chem 101: 1686
Wendelin W, Harler A (1975) Monatsh Chem 106: 1479
Wendelin W, Harler A, Fuchsgruber A (1976) Monatsh Chem 107: 141
Kipping FS (1894) J Chem Soc 65: 498
Witschard G, Griffin CE (1964) J Org Chem 29: 2335
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wendelin, W., Schermanz, K. & Breitmaier, E. Spezielle Reaktionen von α,β-ungesättigten Ketonen, 1. Mitt.: Über die Dimerisierung von 2-Benzyliden-1-indanon in Gegenwart starker Basen. Monatsh Chem 119, 355–365 (1988). https://doi.org/10.1007/BF00810262
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00810262